DEA No. 1670




DEA No. 1670 Brand names, DEA No. 1670 Analogs
DEA No. 1670 Brand Names Mixture
- No information avaliable
DEA No. 1670 Chemical_Formula
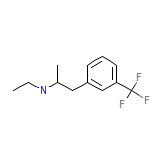
C12H16F3N
DEA No. 1670 RX_link
http://www.rxlist.com/cgi/generic/fenflur.htm
DEA No. 1670 fda sheet
DEA No. 1670 msds (material safety sheet)
DEA No. 1670 Synthesis Reference
No information avaliable
DEA No. 1670 Molecular Weight
231.257 g/mol
DEA No. 1670 Melting Point
108-112 oC at 1.20E+01 mm Hg
DEA No. 1670 H2O Solubility
412 mg/L
DEA No. 1670 State
Liquid
DEA No. 1670 LogP
3.74
DEA No. 1670 Dosage Forms
Capsule (sustained-release); Tablet
DEA No. 1670 Indication
For the management of exogenous obesity as a short-term (a few weeks) adjunct in a regimen of weight reduction based on caloric restriction.
DEA No. 1670 Pharmacology
Used to treat diabetes and obesity, Fenfluramine decreases caloric intake by increasing serotonin levels in the brain's synapses. Fenfluramine acts as a serotonin reuptake inhibitor. It also causes release of serotonin from the synaptosomes.
DEA No. 1670 Absorption
No information avaliable
DEA No. 1670 side effects and Toxicity
No information avaliable
DEA No. 1670 Patient Information
No information avaliable
DEA No. 1670 Organisms Affected
Humans and other mammals