Carlsoma




Carlsoma Brand names, Carlsoma Analogs
- Apesan
- Arusal
- Atonalyt
- Brianil
- Calenfa
- Caprodat
- Carisol
- Carisoma
- Carisoprodate
- Carisoprodatum
- Carlsodol
- Carlsoma
- Carlsoprol
- Carsodal
- Carsodol
- Coprobate
- Diolene
- Domarax
- Flexal
- Flexartal
- Flexartel
- Flibol E
- Isobamate
- Isomeprobamate
- Isopropyl Meprobamate
- Isoprotan
- Isoprotane
- Isoprothane
- Izoprotan
- Mediquil
- Meprobamate
- Meprocon
- Mioartrina
- Miolisodal
- Miolisodol
- Mioratrina
- Mioril
- Mioriodol
- Nospasm
- RELA
- Relasom
- Relax
- Sanoma
- Skutamil
- Soma
- Somadril
- Somalgit
- Somanil
- Stialgin
- Tonolyt Isopropyl Meprobamate
Carlsoma Brand Names Mixture
- No information avaliable
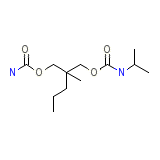
Carlsoma Chemical_Formula
Carlsoma RX_link
Carlsoma fda sheet
Carlsoma msds (material safety sheet)
Carlsoma Synthesis Reference
Carlsoma Molecular Weight
Carlsoma Melting Point
Carlsoma H2O Solubility
Carlsoma State
Carlsoma LogP
Carlsoma Dosage Forms
Carlsoma Indication
Carlsoma Pharmacology
Carlsoma Absorption
Carlsoma side effects and Toxicity
Carlsoma Patient Information
Carisoprodol is a muscle relaxant used to relieve the pain and stiffness of muscle spasms and discomfort due to strain and sprain. Inform your physician if you are pregnant or nursing. Do not take this medication with a monoamine oxidase inhibitor. This medication may cause dizziness, drowsiness, or blurred vision; use caution while driving or operating hazardous machinery. Do not take any other sedating drugs or drink alcohol while taking carisoprodol. If dizziness occurs, avoid sudden changes in posture. Take this medication with food to avoid stomach upset. Notify your physician if you develop trouble breathing, unexplained fever, severe weakness, vision changes, swelling, or skin rash. Withdrawal symptoms may occur if therapy is suddenly stopped in a patient on long-term or high-dose therapy.