Antibiotic Ad 32




Antibiotic Ad 32 Brand names, Antibiotic Ad 32 Analogs
Antibiotic Ad 32 Brand Names Mixture
- No information avaliable
Antibiotic Ad 32 Chemical_Formula
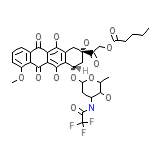
C34H36F3NO13
Antibiotic Ad 32 RX_link
http://www.rxlist.com/cgi/generic2/rubicin.htm
Antibiotic Ad 32 fda sheet
Antibiotic Ad 32 msds (material safety sheet)
Antibiotic Ad 32 Synthesis Reference
No information avaliable
Antibiotic Ad 32 Molecular Weight
723.644 g/mol
Antibiotic Ad 32 Melting Point
116-117oC
Antibiotic Ad 32 H2O Solubility
insoluble
Antibiotic Ad 32 State
Solid
Antibiotic Ad 32 LogP
3.332
Antibiotic Ad 32 Dosage Forms
Liquid
Antibiotic Ad 32 Indication
Bladder cancer
Antibiotic Ad 32 Pharmacology
When 800 mg Valstar was administered intravesically to patients with carcinoma in situ, Valstar penetrated into the bladder wall. The mean total anthracycline concentration measured in bladder tissue exceeded the levels causing 90% cytoxicity to human bladder cells cultured in vitro. During the two-hour dose-retention period, the metabolism of Valstar to its major metabolites N-trifluoroacetyladriamycin and N-trifluoroacetyladriamycinol was neglible. After retention, the drug was almost completely excreted by voiding the instillate. Mean percent recovery of Valstar, N-trifluoroacetyladriamycin, and total anthracyclines in 14 urine samples from six patients was 98.6%, 0.4%, and 99.0% of the total administered drug, respectively. During the two-hour dose-retention period, only nanogram quantities of Valstar were absorbed into the plasma.
Antibiotic Ad 32 Absorption
No information avaliable
Antibiotic Ad 32 side effects and Toxicity
No information avaliable
Antibiotic Ad 32 Patient Information
Antibiotic Ad 32 Organisms Affected
Humans and other mammals