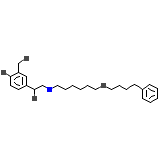
Salmetedur




Categoria
Salmetedur Marchi, Salmetedur Analoghi
Salmetedur Marchi miscela
Salmetedur Formula chimica
C25H37NO4
Salmetedur RX link
http://www.rxlist.com/cgi/generic/salmeterol.htm
Salmetedur FDA foglio
Salmetedur DMS (foglio di materiale di sicurezza)
Salmetedur Sintesi di riferimento
Skidmore SE et al. US Pat. 4.992.474 (1991).
Salmetedur Peso molecolare
415.566 g/mol
Salmetedur Temperatura di fusione
75.5-76.5 oC
Salmetedur H2O Solubilita
Scarsamente solubile
Salmetedur Stato
Solid
Salmetedur LogP
5.022
Salmetedur Forme di dosaggio
Dosato (aerosol); Polvere
Salmetedur Indicazione
Per il trattamento di asma e broncopneumopatia cronica ostruttiva (BPCO).
Salmetedur Farmacologia
Salmeterolo è una lunga durata d'azione beta2-adrenergici agonisti (LABA), di solito solo prescritto per l'asma grave persistente dopo precedente trattamento con una breve durata d'azione beta-agonista come il salbutamolo e è prescritto in concomitanza con un corticosteroide, come beclometasone. La principale differenza notevole di salmeterolo a salbutamolo è che la durata d'azione dura circa 12 ore a confronto con 4-6 ore di salbutamolo. Se usato regolarmente ogni giorno come presecribed, salmeterolo per via inalatoria riduce il numero e la gravità degli attacchi di asma. Tuttavia, non è per l'uso per alleviare un attacco d'asma che ha già iniziato. Salmeterolo per via inalatoria funziona come altri beta 2-agonisti, causando broncodilatazione rilassando la muscolatura liscia delle vie aeree in modo da trattare l'esacerbazione dell'asma. Salmeterolo è simile in azione a formoterolo, tuttavia formoterolo ha dimostrato di avere una maggiore rapidità di azione di salmeterolo in conseguenza di una minore lipofilia, ed è stato anche dimostrato di essere più potente - di 12 mg di dose di formoterolo è stato dimostrato essere equivalente a una dose di 50 mcg di salmeterolo.
Salmetedur Assorbimento
A causa della piccola dose terapeutica, i livelli sistemici di salmeterolo sono bassi o non rilevabili dopo inalazione di dosi raccomandate.
Salmetedur Tossicita
Sintomi di sovradosaggio includono angina (dolore al petto), vertigini, secchezza delle fauci, affaticamento, sintomi simil-influenzali, mal di testa, irregolarità cardiaca, pressione sanguigna alta o bassa, glicemia alta, insonnia, crampi muscolari, nausea, nervosismo, tachicardia, convulsioni e tremori. Per via orale, non si sono verificati decessi in ratti a 1.000 mg / kg (circa 81.000 volte la dose massima raccomandata inalazione al giorno negli adulti e circa 38.000 volte la dose massima giornaliera raccomandata inalazione in bambini su una base mg/m2).
Salmetedur Informazioni paziente
PATIENT INFORMATION
Patients being treated with SEREVENT DISKUS should receive the following information and instructions. This information is
intended to aid them in the safe and effective use of this medication. It is not a disclosure of all possible adverse or
intended effects.
It is important that patients understand how to use the DISKUS appropriately and how to use SEREVENT DISKUS in relation to
other asthma or COPD medications they are taking. Patients should be given the following information:
1. The action of SEREVENT DISKUS may last up to 12 hours or longer. The recommended dosage (1 inhalation twice daily, morning
and evening) should not be exceeded.
2. Most patients are able to taste or feel a dose delivered from SEREVENT DISKUS. However, whether or not patients are able to
sense delivery of a dose, you should instruct them not to exceed the recommended dose of 1 inhalation twice daily, morning and
evening. You should instruct them to contact you or the pharmacist if they have questions.
3. SEREVENT DISKUS is not meant to relieve acute asthma or COPD symptoms and extra doses should not be used for that purpose.
Acute symptoms should be treated with an inhaled, short-acting bronchodilator (the physician should provide the patient with
such medication and instruct the patient in how it should be used).
4. Patients should not stop therapy with SEREVENT DISKUS for asthma or COPD without physician/provider guidance since symptoms
may worsen after discontinuation.
5. �When used for the treatment of EIB, 1 inhalation of SEREVENT DISKUS should be taken 30 minutes before exercise.
� Additional doses of SEREVENT should not be used for 12 hours.
� Patients who are receiving SEREVENT DISKUS twice daily should not use additional SEREVENT for prevention of EIB.
6. The physician should be notified immediately if any of the following situations occur, which may be a sign of seriously
worsening asthma or COPD:
� Decreasing effectiveness of inhaled, short-acting beta2-agonists
� Need for more inhalations than usual of inhaled, short-acting beta2-agonists
� Significant decrease in PEF or lung function as outlined by the physician
� Use of 4 or more inhalations per day of a short-acting beta2-agonist for 2 or more days consecutively
� Use of more than 1 canister (200 inhalations per canister) of an inhaled, short-acting beta2-agonist in an 8-week period.
7. SEREVENT DISKUS should not be used as a substitute for oral or inhaled corticosteroids. The dosage of these medications should
not be changed and they should not be stopped without consulting the physician, even if the patient feels better after initiating
treatment with SEREVENT DISKUS.
8. Patients should be cautioned regarding adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid
heart rate, tremor, or nervousness.
9. When patients are prescribed SEREVENT DISKUS, other medications for asthma and COPD should be used only as directed by the
physician.
10. SEREVENT DISKUS should not be used with a spacer device.
11. Patients who are pregnant or nursing should contact the physician about the use of SEREVENT DISKUS.
12. Effective and safe use of SEREVENT DISKUS includes an understanding of the way that it should be used:
� Never exhale into the DISKUS.
� Never attempt to take the DISKUS apart.
� Always activate and use the DISKUS in a level, horizontal position.
� Never wash the mouthpiece or any part of the DISKUS. KEEP IT DRY.
� Always keep the DISKUS in a dry place.
� Discard 6 weeks after removal from the moisture-protective foil overwrap pouch or after all blisters have been used (when the
dose indicator reads "0" ), whichever comes first.
13. For the proper use of SEREVENT DISKUS and to attain maximum benefit, the patient should read and follow carefully the Patient's
Instructions for Use accompanying the product.
Patients being treated with SEREVENT DISKUS should receive the following information and instructions. This information is
intended to aid them in the safe and effective use of this medication. It is not a disclosure of all possible adverse or
intended effects.
It is important that patients understand how to use the DISKUS appropriately and how to use SEREVENT DISKUS in relation to
other asthma or COPD medications they are taking. Patients should be given the following information:
1. The action of SEREVENT DISKUS may last up to 12 hours or longer. The recommended dosage (1 inhalation twice daily, morning
and evening) should not be exceeded.
2. Most patients are able to taste or feel a dose delivered from SEREVENT DISKUS. However, whether or not patients are able to
sense delivery of a dose, you should instruct them not to exceed the recommended dose of 1 inhalation twice daily, morning and
evening. You should instruct them to contact you or the pharmacist if they have questions.
3. SEREVENT DISKUS is not meant to relieve acute asthma or COPD symptoms and extra doses should not be used for that purpose.
Acute symptoms should be treated with an inhaled, short-acting bronchodilator (the physician should provide the patient with
such medication and instruct the patient in how it should be used).
4. Patients should not stop therapy with SEREVENT DISKUS for asthma or COPD without physician/provider guidance since symptoms
may worsen after discontinuation.
5. �When used for the treatment of EIB, 1 inhalation of SEREVENT DISKUS should be taken 30 minutes before exercise.
� Additional doses of SEREVENT should not be used for 12 hours.
� Patients who are receiving SEREVENT DISKUS twice daily should not use additional SEREVENT for prevention of EIB.
6. The physician should be notified immediately if any of the following situations occur, which may be a sign of seriously
worsening asthma or COPD:
� Decreasing effectiveness of inhaled, short-acting beta2-agonists
� Need for more inhalations than usual of inhaled, short-acting beta2-agonists
� Significant decrease in PEF or lung function as outlined by the physician
� Use of 4 or more inhalations per day of a short-acting beta2-agonist for 2 or more days consecutively
� Use of more than 1 canister (200 inhalations per canister) of an inhaled, short-acting beta2-agonist in an 8-week period.
7. SEREVENT DISKUS should not be used as a substitute for oral or inhaled corticosteroids. The dosage of these medications should
not be changed and they should not be stopped without consulting the physician, even if the patient feels better after initiating
treatment with SEREVENT DISKUS.
8. Patients should be cautioned regarding adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid
heart rate, tremor, or nervousness.
9. When patients are prescribed SEREVENT DISKUS, other medications for asthma and COPD should be used only as directed by the
physician.
10. SEREVENT DISKUS should not be used with a spacer device.
11. Patients who are pregnant or nursing should contact the physician about the use of SEREVENT DISKUS.
12. Effective and safe use of SEREVENT DISKUS includes an understanding of the way that it should be used:
� Never exhale into the DISKUS.
� Never attempt to take the DISKUS apart.
� Always activate and use the DISKUS in a level, horizontal position.
� Never wash the mouthpiece or any part of the DISKUS. KEEP IT DRY.
� Always keep the DISKUS in a dry place.
� Discard 6 weeks after removal from the moisture-protective foil overwrap pouch or after all blisters have been used (when the
dose indicator reads "0" ), whichever comes first.
13. For the proper use of SEREVENT DISKUS and to attain maximum benefit, the patient should read and follow carefully the Patient's
Instructions for Use accompanying the product.
Salmetedur Atto interessato organismi
Gli esseri umani e altri mammiferi