Rosiglizole




Categoria
Rosiglizole Marchi, Rosiglizole Analoghi
Rosiglizole Marchi miscela
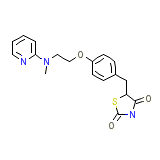
Rosiglizole Formula chimica
Rosiglizole RX link
Rosiglizole FDA foglio
Rosiglizole DMS (foglio di materiale di sicurezza)
Rosiglizole Sintesi di riferimento
Rosiglizole Peso molecolare
Rosiglizole Temperatura di fusione
Rosiglizole H2O Solubilita
Rosiglizole Stato
Rosiglizole LogP
Rosiglizole Forme di dosaggio
Rosiglizole Indicazione
Rosiglizole Farmacologia
Rosiglizole Assorbimento
Rosiglizole Tossicita
Rosiglizole Informazioni paziente
Patients should be informed of the following: Management of type 2 diabetes should include diet control. Caloric restriction, weight loss, and exercise are essential for the proper treatment of the diabetic patient because they help improve insulin sensitivity. This is important not only in the primary treatment of type 2 diabetes, but in maintaining the efficacy of drug therapy.
It is important to adhere to dietary instructions and to regularly have blood glucose and
glycosylated hemoglobin tested. Patients should be advised that it can take 2 weeks to see a reduction in blood glucose and 2 to 3 months to see full effect. Patients should be informed that blood will be drawn to check their liver function prior to the start of therapy and periodically thereafter per the clinical judgement of the healthcare professional. Patients with unexplained symptoms of nausea, vomiting, abdominal pain, fatigue, anorexia, or dark urine should immediately report these symptoms to their physician. Patients who experience an unusually rapid increase in weight or edema or who develop shortness of breath or other symptoms of heart failure while on AVANDIA should immediately report these symptoms to their physician.
AVANDIA can be taken with or without meals.
When using AVANDIA in combination with other hypoglycemic agents, the risk of hypoglycemia, its symptoms and treatment, and conditions that predispose to its development should be explained to patients and their family members.
Therapy with AVANDIA, like other thiazolidinediones, may result in ovulation in some premenopausal anovulatory women. As a result, these patients may be at an increased risk for pregnancy while taking AVANDIA. Thus, adequate contraception in premenopausal women should be recommended. This possible effect has not been specifically investigated in clinical studies so the frequency of this occurrence is not known.
PATIENT INSERT
AVANDIA ® (ah-VAN-dee-a)
Rosiglitazone Maleate Tablets
Read the Patient Information that comes with AVANDIA before you start taking the medicine and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about AVANDIA, ask your doctor or pharmacist.
What is AVANDIA?
AVANDIA is a prescription medicine used with diet and exercise to treat type 2 ("adult onset" or "non-insulin dependent") diabetes mellitus ("high blood sugar"). AVANDIA may be used alone or with other anti-diabetic medicines. AVANDIA can help your body respond better to insulin made in your body. AVANDIA does not cause your body to make more insulin.
Before you take AVANDIA, you should first try to control your diabetes by diet, weight loss, and exercise. In order for AVANDIA to work best, it is very important to exercise, lose excess weight, and follow the diet recommended for your diabetes.
The safety and efficacy of AVANDIA have not been established in children under 18 years of age.
What is Type 2 Diabetes?
Type 2 diabetes happens when a person does not make enough insulin or does not respond normally to the insulin their body makes. When this happens, sugar (glucose) builds up in the blood. This can lead to serious medical problems including kidney damage, heart disease, loss of limbs, and blindness. The main goal of treating diabetes is to lower your blood sugar to a normal level. Lowering and controlling blood sugar may help prevent or delay complications of diabetes such as heart disease, kidney disease or blindness. High blood sugar can be lowered by diet and exercise, by certain medicines taken by mouth, and by insulin shots.
Who should not take AVANDIA?
Do not take AVANDIA if you are allergic to any of the ingredients in AVANDIA. The active ingredient is rosiglitazone maleate. See the end of this leaflet for a list of all the ingredients in AVANDIA.
Before taking AVANDIA, tell your doctor about all your medical conditions, including if you:
•have heart problems or heart failure. AVANDIA can cause your body to keep extra fluid (fluid retention), which leads to swelling and weight gain. Extra body fluid can make some heart problems worse or lead to heart failure.
•have type 1 ("juvenile") diabetes or had diabetic ketoacidosis. These conditions should be treated with insulin.
•have liver problems. Your doctor should do blood tests to check your liver before you start taking AVANDIA and during treatment as needed.
•had liver problems while taking REZULIN ® (troglitazone), another medicine for diabetes.
•are pregnant or trying to become pregnant. It is not known if AVANDIA can harm your unborn baby. You and your doctor should talk about the best way to control your high blood sugar during pregnancy.
•are a premenopausal woman (before the "change of life") who does not have regular monthly periods. AVANDIA may increase your chances of becoming pregnant. Talk to your doctor about birth control choices while taking AVANDIA.
•are breastfeeding. It is not known if AVANDIA passes into breast milk. You should not use AVANDIA while breastfeeding.
•are taking prescription or non-prescription medicines, vitamins or herbal supplements. AVANDIA and certain other medicines can affect each other and lead to serious side effects including high blood sugar or low blood sugar. Keep a list of all the medicines you take. Show this list to your doctor and pharmacist before you start a new medicine. They will tell you if it is okay to take AVANDIA with other medicines.
How should I take AVANDIA?
•Take AVANDIA exactly as prescribed. Your doctor will tell you how many tablets to take and how often. The usual daily starting dose is 4 mg a day taken once a day or 2 mg taken twice a day. Your doctor may need to adjust your dose until your blood sugar is better controlled.
•AVANDIA may be prescribed alone or with other anti-diabetic medicines. This will depend on how well your blood sugar is controlled.
•Take AVANDIA with or without food.
•It can take 2 weeks for AVANDIA to start lowering blood sugar. It may take 2 to 3 months to see the full effect on your blood sugar level.
•If you miss a dose of AVANDIA, take your pill as soon as you remember, unless it is time to take your next dose. Take your next dose at the usual time. Do not take a double dose to make up for a missed dose.
•If you take too much AVANDIA, call your doctor or poison control center right away.
•Test your blood sugar regularly as your doctor tells you.
•Diet and exercise can help your body use its blood sugar better. It is important to stay on your recommended diet, lose excess weight, and get regular exercise while taking AVANDIA.
•Your doctor should do blood tests to check your liver before you start AVANDIA and during treatment as needed. Your doctor should also do regular blood sugar tests (for example, "A1C") to monitor your response to AVANDIA.
What are possible side effects of AVANDIA?
•heart failure. AVANDIA can cause your body to keep extra fluid (fluid retention), which leads to swelling and weight gain. Extra body fluid can make some heart problems worse or lead to heart failure.
•swelling (edema) from fluid retention. Call your doctor right away if you have symptoms such as:
-swelling or fluid retention, especially in the ankles or legs
-shortness of breath or trouble breathing, especially when you lie down
-an unusually fast increase in weight
-unusual tiredness
•low blood sugar (hypoglycemia). Lightheadedness, dizziness, shakiness or hunger may mean that your blood sugar is too low. This can happen if you skip meals, if you use another medicine that lowers blood sugar, or if you have certain medical problems. Call your doctor if low blood sugar levels are a problem for you.
•weight gain. AVANDIA can cause weight gain that may be due to fluid retention or extra body fat. Weight gain can be a serious problem for people with certain conditions including heart problems. Call your doctor if you have an unusually fast increase in weight.
•low red blood cell count (anemia).
•ovulation (release of egg from an ovary in a woman) leading to pregnancy. Ovulation may happen in premenopausal women who do not have regular monthly periods. This can increase the chance of pregnancy.
•liver problems. It is important for your liver to be working normally when you take AVANDIA. Your doctor should do blood tests to check your liver before you start taking AVANDIA and during treatment as needed. Call your doctor right away if you have unexplained symptoms such as:
-nausea or vomiting
-stomach pain
-unusual or unexplained tiredness
-loss of appetite
-dark urine
-yellowing of your skin or the whites of your eyes.
The most common side effects of AVANDIA included cold-like symptoms, injury, and headache.
How should I store AVANDIA?
•Store AVANDIA at room temperature, 59 to 86 °F (15 to 30 °C). Keep AVANDIA in the container it comes in.
•Safely, throw away AVANDIA that is out of date or no longer needed.
•Keep AVANDIA and all medicines out of the reach of children.
General Information about AVANDIA
Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. Do no use AVANDIA for a condition for which it was not prescribed. Do not give AVANDIA to other people, even if they have the same symptoms you have. It may harm them.
This leaflet summarizes important information about AVANDIA. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about AVANDIA that is written for healthcare professionals. You can also find out more about AVANDIA by calling 1-888- 825-5249 or visiting the website www.avandia.com.
What are the ingredients in AVANDIA?
Active Ingredient: rosiglitazone maleate
Inactive Ingredients: hypromellose 2910, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol 3000, sodium starch glycolate, titanium dioxide, triacetin, and 1 or more of the following: synthetic red and yellow iron oxides and talc.
AVANDIA is a registered trademark of GlaxoSmithKline.
REZULIN is a registered trademark of Parke-Davis Pharmaceuticals Ltd.
PIL-AV:L13
©YEAR The GlaxoSmithKline Group of Companies
All rights reserved. Printed in USA. May 2005