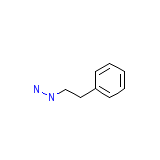
Phenethylhydrazine




Categoria
Phenethylhydrazine Marchi, Phenethylhydrazine Analoghi
Phenethylhydrazine Marchi miscela
Phenethylhydrazine Formula chimica
Phenethylhydrazine RX link
Phenethylhydrazine FDA foglio
Phenethylhydrazine DMS (foglio di materiale di sicurezza)
Phenethylhydrazine Sintesi di riferimento
Phenethylhydrazine Peso molecolare
Phenethylhydrazine Temperatura di fusione
Phenethylhydrazine H2O Solubilita
Phenethylhydrazine Stato
Phenethylhydrazine LogP
Phenethylhydrazine Forme di dosaggio
Phenethylhydrazine Indicazione
Phenethylhydrazine Farmacologia
Phenethylhydrazine Assorbimento
Phenethylhydrazine Tossicita
Phenethylhydrazine Informazioni paziente
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with NARDIL and should counsel them in its appropriate use. A patient Medication Guide About Using Antidepressants in Children and Teenagers is available for NARDIL. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking NARDIL.
Clinical Worsening and Suicide Risk: Patients, their families, and their caregivers should be encouraged to be alert to the emergence of anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, mania, other unusual changes in behavior, worsening of depression, and suicidal ideation, especially early during antidepressant treatment and when the dose is adjusted up or down. Families and caregivers of patients should be advised to observe for the emergence of such symptoms on a day-to-day basis, since changes may be abrupt. Such symptoms should be reported to the patient's prescriber or health professional, especially if they are severe, abrupt in onset, or were not part of the patient's presenting symptoms. Symptoms such as these may be associated with an increased risk for suicidal thinking and behavior and indicate a need for very close monitoring and possibly changes in medication.
MEDICATION GUIDE
About Using Antidepressants in Children and Teenagers
What is the most important information I should know if my child is being prescribed an antidepressant?
Parents or guardians need to think about 4 important things when their child is prescribed an antidepressant:
- There is a risk of suicidal thoughts or actions
- How to try to prevent suicidal thoughts or actions in your child
- You should watch for certain signs if your child is taking an antidepressant
- There are benefits and risks when using antidepressants
1. There is a Risk of Suicidal Thoughts or Actions
Children and teenagers sometimes think about suicide, and many report trying to kill themselves.
Antidepressants increase suicidal thoughts and actions in some children and teenagers. But suicidal thoughts and actions can also be caused by depression, a serious medical condition that is commonly treated with antidepressants. Thinking about killing yourself or trying to kill yourself is called suicidality or being suicidal.
A large study combined the results of 24 different studies of children and teenagers with depression or other illnesses. In these studies, patients took either a placebo (sugar pill) or an antidepressant for 1 to 4 months. No one committed suicide in these studies, but some patients became suicidal. On sugar pills, 2 out of every 100 became suicidal. On the antidepressants, 4 out of every 100 patients became suicidal.
For some children and teenagers, the risks of suicidal actions may be especially high.These include patients with
- Bipolar illness (sometimes called manic-depressive illness)
- A family history of bipolar illness
- A personal or family history of attempting suicide
If any of these are present, make sure you tell your health care provider before your child takes an antidepressant.
2. How to Try to Prevent Suicidal Thoughts and Actions
To try to prevent suicidal thoughts and actions in your child, pay close attention to changes in her or his moods or actions, especially if the changes occur suddenly. Other important people in your child's life can help by paying attention as well (e.g., your child, brothers and sisters, teachers, and other important people). The changes to look out for are listed in Section 3, on what to watch for.
Whenever an antidepressant is started or its dose is changed, pay close attention to your child.
After starting an antidepressant, your child should generally see his or her health care provider
- Once a week for the first 4 weeks
- Every 2 weeks for the next 4 weeks
- After taking the antidepressant for 12 weeks
- After 12 weeks, follow your health care provider's advice about how often to come back
- More often if problems or questions arise
You should call your child's health care provider between visits if needed.
3. You Should Watch for Certain Signs If Your Child is Taking an Antidepressant
Contact your child's health care provider right away if your child exhibits any of the following signs for the first time, or if they seem worse, or worry you, your child, or your child's teacher:
- Thoughts about suicide or dying
- Attempts to commit suicide
- New or worse depression
- New or worse anxiety
- Feeling very agitated or restless
- Panic attacks
- Difficulty sleeping (insomnia)
- New or worse irritability
- Acting aggressive, being angry, or violent
- Acting on dangerous impulses
- An extreme increase in activity and talking
- Other unusual changes in behavior or mood
Never let your child stop taking an antidepressant without first talking to his or her health care provider. Stopping an antidepressant suddenly can cause other symptoms.
Prozac™ is a registered trademark of Eli Lilly and Company.
Zoloft™ is a registered trademark of Pfizer Pharmaceuticals.
Anafranil™ is a registered trademark of Mallinckrodt Inc.
This Medication Guide has been approved by the US Food and Drug Administration for all antidepressants.