Mometasone furoate




Categorie
Mometasone furoate Les marques, Mometasone furoate Analogs
Mometasone furoate Les marques melange
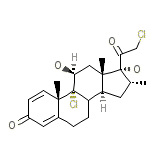
Mometasone furoate Formule chimique
Mometasone furoate RX lien
Mometasone furoate FDA fiche
Mometasone furoate msds (fiche de securite des materiaux)
Mometasone furoate Synthese de reference
Mometasone furoate Poids moleculaire
Mometasone furoate Point de fusion
Mometasone furoate H2O Solubilite
Mometasone furoate Etat
Mometasone furoate LogP
Mometasone furoate Formes pharmaceutiques
Mometasone furoate Indication
Mometasone furoate Pharmacologie
Mometasone furoate Absorption
Mometasone furoate Toxicite
Mometasone furoate Information pour les patients
PATIENT'S INSTRUCTIONS FOR USE ASMANEX® TWISTHALER® 220 mcg (mometasone furoate inhalation powder)
FOR ORAL INHALATION ONLY
Please read this leaflet carefully before taking ASMANEX® TWISTHALER®. This leaflet does not contain the complete information about this medication. If you have any questions about ASMANEX® TWISTHALER®, ask your healthcare provider or pharmacist.
IMPORTANT POINTS TO REMEMBER ABOUT ASMANEX® TWISTHALER®
• Your healthcare provider has prescribed ASMANEX® TWISTHALER®. It contains a medication called mometasone furoate, which is a synthetic corticosteroid. This medication is used as maintenance treatment that helps prevent and control asthma symptoms.
• ASMANEX® TWISTHALER® is not a bronchodilator. You should not use ASMANEX® TWISTHALER® when you are having sudden symptoms of shortness of breath. Use an inhaled short-acting bronchodilator such as albuterol to relieve sudden symptoms of shortness of breath.
• Your healthcare provider may prescribe bronchodilators such as albuterol for emergency relief if an acute asthma attack occurs.
• The inhaler delivers your medication as a very fine powder that you may (b)(4) mell or feel. Whether or not you are able to sense of a dose, not take extra ess your healthcare provider has told you to.
• Use the inhaler regularly and at the same time each day as prescribed by your healthcare provider. Maximum benefit may not be achieved for 1 to 2 weeks or longer. If your symptoms do not improve in that time frame or if your condition worsens, contact your healthcare provider.
Do not use the inhaler if you notice that it is not working correctly. Take it to your healthcare provider or pharmacist.
HOW TO USE
Remove the ASMANEX® TWISTHALER® from its foil pouch and write the date on the cap label. It's important to throw away the inhaler 45 days after this date or when the dose counter reads "00", whichever comes first.
Step 1. Open inhaler
Hold the inhaler straight up with the pink portion (the base) on the bottom. It is important that you remove the cap of the TWISTHALER® while it is in this upright position to make sure that you get the right amount of medicine with each dose.
Holding the pink base, twist the cap in a counter- clockwise direction to remove it. As you lift off the cap, the dose counter on the base will count down by one. (If you began with the dose counter reading "30," this action will cause it to now read "29.") This action loads the device with the medicine that you are now ready to inhale.
IT IS IMPORTANT TO NOTE that the indented arrow (located on the white portion of the TWISTHALER®, directly above the pink base) is pointing to the dose counter.
Step 2. Inhale dose
Exhale fully. Then bring the TWISTHALER® up to your mouth with the mouthpiece facing toward you. Place it in your mouth, holding it in a horizontal position. Firmly closing your lips around the mouthpiece, take in a fast, deep breath. Since the medication is a very fine powder, you may not be able to feel or taste it after inhalation.
Remove the TWISTHALER® from your mouth and hold your breath for about 10 seconds, or as long as you comfortably can.
IMPORTANT: DO NOT BREATHE OUT THROUGH THE INHALER.
After you take your medicine, it is important that you wipe the mouthpiece dry, if necessary, and immediately replace the cap, firmly closing the TWISTHALER®.
This is the only way to be sure that your next dose is properly loaded. Be sure that the arrow is in line with the dose-counter window. The cap needs to be put back on and turned in a clockwise direction, as you gently press down. You'll hear a distinctive "click" to let you know that the cap is fully closed.
IT IS IMPORTANT TO REPEAT STEPS 1 AND 2 EACH TIME YOU INHALE.
Rinse your mouth after using.
STORING YOUR INHALER
Keep your inhaler clean and dry at all times. If the device needs cleaning, gently wipe the mouthpiece with a dry cloth or tissue as needed. Do not wash the inhaler. Avoid contact with any liquids.
Store in a dry place at 25°C (77F°); excursions permitted to 15-30°C (59-86°F)
Keep your inhaler out of the reach of young children.
HOW TO KNOW WHEN YOUR INHALER IS EMPTY
The inhaler has a dose indicator window on the pink base. It is a dose counter which displays the number of doses remaining. When the unit reads 01, this indicates the last remaining dose. After dose 01, the counter will read 00, and the cap will lock. The unit must then be discarded.