Mitoxantrone hydrochloride




Categorie
Mitoxantrone hydrochloride Les marques, Mitoxantrone hydrochloride Analogs
Mitoxantrone hydrochloride Les marques melange
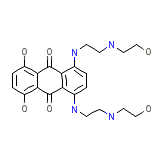
Mitoxantrone hydrochloride Formule chimique
Mitoxantrone hydrochloride RX lien
Mitoxantrone hydrochloride FDA fiche
Mitoxantrone hydrochloride msds (fiche de securite des materiaux)
Mitoxantrone hydrochloride Synthese de reference
Mitoxantrone hydrochloride Poids moleculaire
Mitoxantrone hydrochloride Point de fusion
Mitoxantrone hydrochloride H2O Solubilite
Mitoxantrone hydrochloride Etat
Mitoxantrone hydrochloride LogP
Mitoxantrone hydrochloride Formes pharmaceutiques
Mitoxantrone hydrochloride Indication
Mitoxantrone hydrochloride Pharmacologie
Mitoxantrone hydrochloride Absorption
Mitoxantrone hydrochloride Toxicite
Mitoxantrone hydrochloride Information pour les patients
NOVANTRONEâ may impart a blue-green color to the urine for 24 hours after administration, and patients should be advised to expect this during therapy. Bluish discoloration of the sclera may also occur. Patients should be advised of the signs and symptoms of myelosuppression.
Patients with multiple sclerosis should be provided with the Patient Package Insert at the time that the decision is made to treat with NOVANTRONEâ and prior to and in close temporal proximity to each treatment. In addition, the physician should discuss the issues addressed in the Patient Package Insert with the patient.
Patient Package Insert
NOVANTRONE (noe-VAN-trone)
mitoxantrone for injection concentrate
SERONO
For Treating Multiple Sc1erosis
Read this information carefully before you start taking NOVOVANTRONE for multiple sclerosis (MS). This information does not take the place of talking with your doctor. Your doctor can tell you more about NOVANTRONE and answer any questions you have about this treatment.
NOVANTRONE is used for other conditions besides MS. This 1eaflet has information about using NOVANTRONE specifically for MS.
What is the most important in formation I should know about NOVANTRONE?
NOVANTRONE can reduce relapses and disability for patients with worsening forms of MS.
- NOVANTRONE may damage your heart at any time during therapy or months to years after therapy ends. Heart damage caused by NOVANTRONE can be serious and may cause death. Your doctor will perform certain tests to see that your heart is working normally before you start to take NOVANTRONE. Your doctor will repeat these heart tests before you receive each additional dose. Your doctor wi11 also perform these tests if you have any symptoms of heart problems. Because the risk to your heart may depend on the total amount of NOVANTRONE given, your doctor will limit the number of doses you get. Most patients will reach this limit after about 8 to 12 doses given over 2 to 3 years. After you have reached your limit, you should not receive any additional NOVANTRONE. You and your doctor should both keep track of how much NOVANTRONE you get.
NOVANTRONE can increase your chance of getting an infection. If you begin to have any signs of infection, such as fever, chills, sore throat, cough, pain with urinating, or urinating more often, call your doctor right away. If you have such an infection, it can usually be treated by taking antibiotics.
MS and cancer patients treated with NOVANTRONE have an increased risk of developing leukemia.
What is NOVANTRONE?
NOVANTRONE is a medicine to treat MS patients with secondary (chronic) progressive, progressing relapsing, or worsening relapsing-remitting MS. It is not for treating primary progressive MS. Patients treated with NOVANTRONE may have fewer relapses and keep their mobility longer.
Who should not take NOVANTRONE?
Women who are pregnant, are trying to become pregnant, or are breastfeeding should not take NOVANTRONE because it may harm the baby. You should use birth control while taking NOVANTRONE to avoid becoming pregnant. Your doctor should also give you a pregnancy test before each dose, and you should know the results of this test before you get each dose of NOVANTRONE. If you plan on getting pregnant, talk with your doctor about stopping the NOVANTRONE treatments. If you do become pregnant, contact your doctor right away.
You should not take NOVANTRONE if your doctor finds you have a low number of white blood cells (leukocytes).
You should not take NOVANTRONE if our doctor finds heartís ability to pump blood is decreased.
If you are allergic to NOVANTRONE, you should not take it. The active ingredient is mitoxantrone. Ask your doctor about the inactive ingredients.
Your doctor needs to know the following information about you to help decide if NOVANTRONE is right for you. Tell your doctor if you have now or had in the past
- heart diseases
- treatment with NOVANTRONE
- cancer chemotherapy treatment
- radiation treatment to the chest area
- blood-clotting problems
- anemia or low red blood cell counts
- low white blood cell counts
- unusual or unexpected bleeding
- infections
- liver disease or problems
- any known allergies or sensitivities
Also tell your doctor if you take other medicines, including nonprescription medicines and nutritional supplements.
How do I take NOVANTRONE?
NOVANTRONE is given through a needle placed in a vein in your arm. The dose takes about 5 to 15 minutes to deliver. NOVANTRONE treatment is usually given once every 3 months for about 2 to 3 years (8 to 12 doses). However, this may differ for different patients.
What diagnostic tests will be performed?
You will need to have regular testing of your heart and blood to help avoid serious side; effects.
Before each dose of NOVANTRONE, your doctor will take blood samples to check your blood counts and liver function. Your doctor may also take a blood sample if you begin to have signs of an infection. If you are a woman who is capable of becoming pregnant, even if you were using birth control, you must have a pregnancy test before each NOVANTRONE dose, and you should know the results before you receive each NOVANTRONE dose.
To measure possible changes to the heart, you should have regular testing of your heartís ability to pump blood. This requires taking pictures of your heart using a simple, painless test such as an echocardiogram. Your heart should be tested before each dose of NOVANTRONE, or if you show signs of heart problems.
You and your doctor should carefully track the total amount of NOVANTRONE you get. Your doctor may stop NOVANTRONE if your tests show that your heartís ability to pump blood has decreased. If you change doctors, make sure your new doctor knows how much NOVANTRONE you have taken.
What should I avoid while taking NOVANTRONE?
Women should not become pregnant or breastfeed while taking NOVANTRONE because it may harm the baby. Talk with your doctor about effective birth control. Tell your doctor if you become pregnant.
Talk with your doctor about any medicines you currently take and any medicines you plan to start or stop taking. These include prescription and non-prescription medicines and nutritional supplements. Some medicines may affect how NOVANTRONE works.
What are the possible side effects of NOVANTRONE?
Most side effects of NOVANTRONE are not severe and can normally be treated by your doctor. The most common side effects of NOVANTRONE in patients with MS are nausea, hair thinning, loss of menstrual periods, bladder infections, and mouth sores. The nausea is usually mild and generally lasts for less than 24 hours. A small number of patients treated with NOVANTRONE develop heart problems. Tell your doctor if you have trouble breathing, swelling of your legs or ankles, or uneven or fast heartbeat.
NOVANTRONE may cause your white blood cell count to go down, which increases your chance of getting an infection. This risk is greatest within one month after each dose. In addition, NOVANTRONE may cause your platelet count to go down, which increases your chance of bleeding. Cal] your doctor right away if you begin to have fever, chills, sore throat, cough, pain with urination, urination more often, or if you notice any unusual bleeding or bruising.
NOVANTRONE is dark blue in color, so it may turn your urine a blue-green color for a few days after each dose. The white part of your eyes may also have a slight blue color.
Other side effects may occur be sure to tell your doctor about any side effects whether or not they are listed here.
General advice about prescription medicines
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. If you have any concerns about NOVANTRONE, ask your doctor. Your doctor can give you information about NOVANTRONE that was written for health care professionals. For more information call MS LifeLines toll free at 1-877-447-3243.
Manufactured for:
Serono, Inc.
Rockland, MA 02370, USA
PI 7834-3
Issued 4/2005