Kenacort-A




Categorie
Kenacort-A Les marques, Kenacort-A Analogs
- Adcortyl
- Aristocort
- Aristocort A
- Aristocort Tablets
- Aristogel
- Aristospan
- Azmacort
- Celeste
- Cinolone
- Cinolone-T
- Delphicort
- Fluoxiprednisolone
- Fluoxyprednisolone
- Flutex
- Kenacort
- Kenacort-A
- Kenacort-Ag
- Kenalog
- Kenalog in Orabase
- Kenalog-10
- Kenalog-40
- Kenalog-H
- Ledercort
- Nasacort
- Nasacort Aq
- Nasacort Hfa
- Omcilon
- Omicilon
- Oracort
- Oralone
- Orion
- Polcortolon
- Rodinolone
- Sk-Triamcinolone
- Tiamcinolonum [Inn-Latin]
- Tri-Nasal
- Triacet
- Triacort
- Triam-Tablinen
- Triamcet
- Triamcinalone
- Triamcinlon
- Triamcinolon
- Triamcinolona [Inn-Spanish]
- Triamcinolone Acetonide
- Triamcinolone Acetonide in Absorbase
- Triamcinolone Diacetate
- Triamcinolone [Usan:Ban:Inn:Jan]
- Triamcinolonum [Inn]
- Triatex
- Tricortale
- Triderm
- Trymex
- Vetalog
- Volon
Kenacort-A Les marques melange
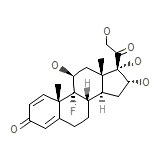
Kenacort-A Formule chimique
Kenacort-A RX lien
Kenacort-A FDA fiche
Kenacort-A msds (fiche de securite des materiaux)
Kenacort-A Synthese de reference
Kenacort-A Poids moleculaire
Kenacort-A Point de fusion
Kenacort-A H2O Solubilite
Kenacort-A Etat
Kenacort-A LogP
Kenacort-A Formes pharmaceutiques
Kenacort-A Indication
Kenacort-A Pharmacologie
Kenacort-A Absorption
Kenacort-A Toxicite
Kenacort-A Information pour les patients
Patients being treated with Azmacort Inhalation Aerosol should receive the following information and instructions. This information is intended to aid them in the safe and effective use of this medication. It is not a complete disclosure of all possible adverse or intended effects.
Patients should use Azmacort Inhalation Aerosol at regular intervals as directed. Results of clinical trials indicate that significant improvement in asthma may occur by 1 week, but maximum benefit may not be achieved for 2 weeks or more. The patient should not increase the prescribed dosage but should contact the physician if symptoms do not improve or if the condition worsens.
In clinical studies and post-marketing experience with Azmacort Inhalation Aerosol, local infections of the oropharynx with Candida albicans have occurred. When such an infection develops, it should be treated with appropriate local or systemic (i.e., oral antifungal) therapy while remaining on treatment with Azmacort Inhalation Aerosol. However, at times therapy with Azmacort Inhalation Aerosol may need to be interrupted.
Patients should be instructed to track their use of Azmacort Inhalation Aerosol and to dispose of the canister after 240 actuations since reliable dose delivery cannot be assured after 240 doses.
Patients who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles and if exposed, to obtain medical advice.