Diphenoxylate-Atropine




Categorie
Diphenoxylate-Atropine Les marques, Diphenoxylate-Atropine Analogs
Diphenoxylate-Atropine Les marques melange
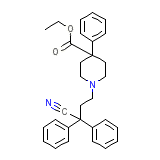
Diphenoxylate-Atropine Formule chimique
Diphenoxylate-Atropine RX lien
Diphenoxylate-Atropine FDA fiche
Diphenoxylate-Atropine msds (fiche de securite des materiaux)
Diphenoxylate-Atropine Synthese de reference
Diphenoxylate-Atropine Poids moleculaire
Diphenoxylate-Atropine Point de fusion
Diphenoxylate-Atropine H2O Solubilite
Diphenoxylate-Atropine Etat
Diphenoxylate-Atropine LogP
Diphenoxylate-Atropine Formes pharmaceutiques
Diphenoxylate-Atropine Indication
Diphenoxylate-Atropine Pharmacologie
Diphenoxylate-Atropine Absorption
Diphenoxylate-Atropine Toxicite
Diphenoxylate-Atropine Information pour les patients
INFORM THE PATIENT (PARENT OR GUARDIAN) NOT TO EXCEED THE RECOMMENDED DOSAGE AND TO KEEP DIPHEN-OXYLATE HCL AND ATROPINE SULFATE OUT OF THE REACH OF CHILDREN AND IN A CHILD-RESISTANT CONTAINER, INFORM THE PATIENT OF THE CONSEQUENCES OF OVERDOSAGE, INCLUDING SEVERE RESPIRATORY DEPRESSION AND COMA, POSSIBLY LEADING TO PERMANENT BRAIN DAMAGE OR DEATH.
Diphenoxylate HCl and atropine sulfate may produce drowsiness or dizziness. The patient should be cautioned regarding activities requiring mental alertness, such as driving or operati ng dangerous machinery. Potentiation of the action of alcohol, barbiturates and tranquilizers with concomitant use of diphenoxylate HCl and atropine sulfate should be explained to the patient. The physician should also provide the patient with other information in this labeling, as appropriate.