Alotec




Categorie
Alotec Les marques, Alotec Analogs
Alotec Les marques melange
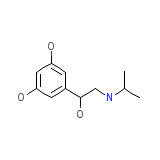
Alotec Formule chimique
C11H17NO3
Alotec RX lien
http://www.rxlist.com/cgi/generic2/metaproterenol.htm
Alotec FDA fiche
Alotec msds (fiche de securite des materiaux)
Alotec Synthese de reference
O. Thoma, K. Zeile, le brevet américain. 3,341,594 (1967)
Alotec Poids moleculaire
211.258 g/mol
Alotec Point de fusion
100oC
Alotec H2O Solubilite
9.7mg / L
Alotec Etat
Solid
Alotec LogP
1.172
Alotec Formes pharmaceutiques
Inhalation
Alotec Indication
Pour le traitement du bronchospasme, bronchite chronique, l'asthme et l'emphysème.
Alotec Pharmacologie
Metaproterenol, une amine synthétique, est structurellement et pharmacologiquement analogue à l'isoprotérénol. Metaproterenol est utilisé exclusivement comme un bronchodilatateur. Les effets pharmacologiques des agonistes bêta-adrénergiques médicaments, tels que Metaproterenol, sont au moins en partie attribuable à la stimulation par le biais des récepteurs bêta-adrénergiques adénylcyclase intracellulaire, l'enzyme qui catalyse la conversion de l'adénosine triphosphate (ATP) pour cyclique-3 ', 5'-adénosine monophosphate (AMP-c). Augmentation de c-AMP niveaux sont associées à la relaxation des muscles lisses bronchiques et l'inhibition de la libération de médiateurs de l'immédiate hypersensibilité à partir des cellules, en particulier par les mastocytes.
Alotec Absorption
3% (biodisponibilité orale de 40%)
Alotec Toxicite
Les symptômes de surdose comprennent l'angine de poitrine, hypertension ou hypotension, arythmies, nervosité, maux de tête, tremblements, bouche sèche, palpitations, nausées, étourdissements, fatigue, malaise et d'insomnie. DL50 = 42 mg / kg (par voie orale chez le rat).
Alotec Information pour les patients
Patient�s Instructions for Use Alupent�
(metaproterenol sulfate USP) Inhalation Aerosol
1. Insert metal canister into clear end of mouthpiece.
2. Remove protective cap, invert canister and shake well before each use.
3. Avoid spraying in eyes.
4. Enclose mouthpiece with the lips. The base of the canister should be held vertically. The Alupent� canister is to be used only with the white Alupent� Inhalation Aerosol mouthpiece. This mouthpiece should not be used with other aerosol medications.
5. Exhale deeply, then inhale slowly through the mouth and at the same time firmly press once on the upended canister base; continue to inhale deeply. Hold your breath for a few seconds and then remove the mouthpiece from the mouth and exhale slowly.
One inhalation is often enough to obtain relief. The inhalation can be repeated once or twice, if necessary, or as your physician directs. Wait at least two minutes before repeating the inhalation. In most cases, the dose should not be repeated more often than every 3 to 4 hours. No more than 12 inhalations should be taken in one day.
6. Replace protective cap after use.
WARNING
Do not exceed the dose prescribed by your physician. If difficulty in breathing persists, contact your physician immediately.
Note: When full, the container holds enough medication for at least 200 inhalations: at least 100 inhalations are in the sample unit. Check regularly, by shaking the cylinder or container, to determine whether it contains any medication. When it first seems empty, there are still about ten doses left. Refill containers for the plastic mouthpiece are available when prescribed by your physician.
Keep the mouthpiece clean. Wash with hot water. If soap is used, rinse thoroughly with plain water.
Never open the container holding the medication. Opening it is dangerous and renders the contents useless.
Caution: Contents Under Pressure. Do not puncture or incinerate container. Do not expose to heat or store at temperatures above 120�F. Keep out of reach of small children
Note: The indented statement below is required by the Federal government�s Clean Air Act for all products containing or manufactured with chlorofluorocarbons (CFCs): This product contains trichloromonofluoromethane (CFC-11), dichlorodifluoromethane (CFC-12) and dichlorotetrafluoroethane (CFC-114), substances which harm the environment by destroying ozone in the upper atmosphere.
Your physician has determined that this product is likely to help your personal health. USE THIS PRODUCT AS DIRECTED, UNLESS INSTRUCTED TO DO OTHERWISE BY YOUR PHYSICIAN. If you have any questions about alternatives, consult with your physician
(metaproterenol sulfate USP) Inhalation Aerosol
1. Insert metal canister into clear end of mouthpiece.
2. Remove protective cap, invert canister and shake well before each use.
3. Avoid spraying in eyes.
4. Enclose mouthpiece with the lips. The base of the canister should be held vertically. The Alupent� canister is to be used only with the white Alupent� Inhalation Aerosol mouthpiece. This mouthpiece should not be used with other aerosol medications.
5. Exhale deeply, then inhale slowly through the mouth and at the same time firmly press once on the upended canister base; continue to inhale deeply. Hold your breath for a few seconds and then remove the mouthpiece from the mouth and exhale slowly.
One inhalation is often enough to obtain relief. The inhalation can be repeated once or twice, if necessary, or as your physician directs. Wait at least two minutes before repeating the inhalation. In most cases, the dose should not be repeated more often than every 3 to 4 hours. No more than 12 inhalations should be taken in one day.
6. Replace protective cap after use.
WARNING
Do not exceed the dose prescribed by your physician. If difficulty in breathing persists, contact your physician immediately.
Note: When full, the container holds enough medication for at least 200 inhalations: at least 100 inhalations are in the sample unit. Check regularly, by shaking the cylinder or container, to determine whether it contains any medication. When it first seems empty, there are still about ten doses left. Refill containers for the plastic mouthpiece are available when prescribed by your physician.
Keep the mouthpiece clean. Wash with hot water. If soap is used, rinse thoroughly with plain water.
Never open the container holding the medication. Opening it is dangerous and renders the contents useless.
Caution: Contents Under Pressure. Do not puncture or incinerate container. Do not expose to heat or store at temperatures above 120�F. Keep out of reach of small children
Note: The indented statement below is required by the Federal government�s Clean Air Act for all products containing or manufactured with chlorofluorocarbons (CFCs): This product contains trichloromonofluoromethane (CFC-11), dichlorodifluoromethane (CFC-12) and dichlorotetrafluoroethane (CFC-114), substances which harm the environment by destroying ozone in the upper atmosphere.
Your physician has determined that this product is likely to help your personal health. USE THIS PRODUCT AS DIRECTED, UNLESS INSTRUCTED TO DO OTHERWISE BY YOUR PHYSICIAN. If you have any questions about alternatives, consult with your physician
Alotec Organismes affectes
Les humains et autres mammifères