Tabrin




Categoria
Tabrin Nombres de marca, Tabrin Analogos
- Akilen
- Baccidal
- Bactocin
- Danoflox
- Effexin
- Exocin
- Exocine
- Flobacin
- Flodemex
- Flotavid
- Flovid
- Floxal
- Floxil
- Floxin
- Floxin Otic
- Floxstat
- Fugacin
- Inoflox
- Kinflocin
- Kinoxacin
- Liflox
- Loxinter
- Marfloxacin
- Medofloxine
- Mergexin
- Novecin
- Nufafloqo
- O-Flox
- Obide
- Occidal
- Ocuflox
- Ofcin
- Oflin
- Oflocee
- Oflocet
- Oflocin
- Oflodal
- Oflodex
- Oflodura
- Oflox
- Ofloxin
- Ofus
- Onexacin
- Operan
- Orocin
- Otonil
- Pharflox
- Praxin
- Puiritol
- Qinolon
- Qipro
- Quinolon
- Quotavil
- Rilox
- Sinflo
- Tabrin
- Taravid
- Tariflox
- Tarivid
- Tarivid Eye Ear
- Tarivid Otic
- Telbit
- Tructum
- Uro Tarivid
- Viotisone
- Zanocin
Tabrin Marca los nombres de mezcla
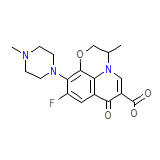
Tabrin Formula quimica
C18H20FN3O4
Tabrin RX enlace
http://www.rxlist.com/cgi/generic/oflox.htm
Tabrin FDA hoja
Tabrin MSDS (hoja de seguridad de materiales)
Tabrin Sintesis de referencia
I. Hayakava et al., Patente de EE.UU.. 4,382,892 (1983)
Tabrin Peso molecular
361.368 g/mol
Tabrin Punto de fusion
250-257 oC
Tabrin H2O Solubilidad
28,3 mg / ml
Tabrin Estado
Solid
Tabrin LogP
1.268
Tabrin Formas de dosificacion
Solución (inyección IV), Tablet, gotas oftálmicas
Tabrin Indicacion
Para el tratamiento de las infecciones (tracto respiratorio, riñón, piel, tejidos blandos, infección del tracto urinario), la uretra y la gonorrea cervical.
Tabrin Farmacologia
Ofloxacino es un antibiótico de quinolona / fluoroquinolonas. Ofloxacina es bactericida y su mecanismo de acción depende de bloqueo de la replicación del ADN bacteriano mediante la unión en sí a una enzima llamada ADN girasa, que permite que el destrenzado necesarios para reproducir una doble hélice de ADN en dos. Cabe destacar que la droga tiene una afinidad 100 veces mayor de la ADN girasa bacteriana que en los mamíferos. Ofloxacino es un amplio espectro antibiótico que es activo frente a bacterias Gram-positivas y Gram-negativas.
Tabrin Absorcion
La biodisponibilidad de ofloxacina en la formulación del comprimido es de aproximadamente el 98%
Tabrin Toxicidad
DL50 = 5450 mg / kg (por vía oral en ratones)
Tabrin Informacion de Pacientes
PATIENT INFORMATION
Patients should be advised:
- to drink fluids liberally;
- that mineral supplements, vitamins with iron or minerals, calcium- , aluminum-, or magnesium-based
antacids, sucralfate or Videx�, (Didanosine), chewable/buffered tablets or the pediatric powder for
oral solution should not be taken within the two-hour period before or within the two-hour period after
taking ofloxacin (See Drug Interactions);
- that ofloxacin can be taken without regard to meals;
- that ofloxacin may cause neurologic adverse effects (e. g. , dizziness, lightheadedness) and that
patients should know how they react to ofloxacin before they operate an automobile or machinery or
engage in activities requiring mental alertness and coordination
- to discontinue treatment and inform their physician if they experience pain, inflammation, or rupture
of a tendon, and to rest and refrain from exercise until the diagnosis of tendinitis or tendon rupture
has been confidently excluded;
- that ofloxacin may be associated with hypersensitivity reactions, even following the first dose, to
discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat,
difficulty in swallowing or breathing, any swelling suggesting angioedema (e. g. , swelling of the lips,
tongue, face; tightness of the throat, hoarseness), or any other symptom of an allergic reaction
- to avoid excessive sunlight or artificial ultraviolet light while receiving ofloxacin and to discontinue
therapy if phototoxicity (e. g. , skin eruption) occurs;
- that if they are diabetic and are being treated with insulin or an oral hypoglycemic drug, to discontinue
ofloxacin immediately if a hypoglycemic reaction occurs and consult a physician
- that convulsions have been reported in patients taking quinolones, including ofloxacin, and to notify their
physician before taking this drug if there is a history of this condition.
Patients should be advised:
- to drink fluids liberally;
- that mineral supplements, vitamins with iron or minerals, calcium- , aluminum-, or magnesium-based
antacids, sucralfate or Videx�, (Didanosine), chewable/buffered tablets or the pediatric powder for
oral solution should not be taken within the two-hour period before or within the two-hour period after
taking ofloxacin (See Drug Interactions);
- that ofloxacin can be taken without regard to meals;
- that ofloxacin may cause neurologic adverse effects (e. g. , dizziness, lightheadedness) and that
patients should know how they react to ofloxacin before they operate an automobile or machinery or
engage in activities requiring mental alertness and coordination
- to discontinue treatment and inform their physician if they experience pain, inflammation, or rupture
of a tendon, and to rest and refrain from exercise until the diagnosis of tendinitis or tendon rupture
has been confidently excluded;
- that ofloxacin may be associated with hypersensitivity reactions, even following the first dose, to
discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat,
difficulty in swallowing or breathing, any swelling suggesting angioedema (e. g. , swelling of the lips,
tongue, face; tightness of the throat, hoarseness), or any other symptom of an allergic reaction
- to avoid excessive sunlight or artificial ultraviolet light while receiving ofloxacin and to discontinue
therapy if phototoxicity (e. g. , skin eruption) occurs;
- that if they are diabetic and are being treated with insulin or an oral hypoglycemic drug, to discontinue
ofloxacin immediately if a hypoglycemic reaction occurs and consult a physician
- that convulsions have been reported in patients taking quinolones, including ofloxacin, and to notify their
physician before taking this drug if there is a history of this condition.
Tabrin Organismos afectados
Bacterias entéricas y otras eubacterias