Stocrin




Categoria
Stocrin Nombres de marca, Stocrin Analogos
Stocrin Marca los nombres de mezcla
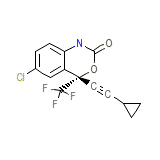
Stocrin Formula quimica
Stocrin RX enlace
Stocrin FDA hoja
Stocrin MSDS (hoja de seguridad de materiales)
Stocrin Sintesis de referencia
Stocrin Peso molecular
Stocrin Punto de fusion
Stocrin H2O Solubilidad
Stocrin Estado
Stocrin LogP
Stocrin Formas de dosificacion
Stocrin Indicacion
Stocrin Farmacologia
Stocrin Absorcion
Stocrin Toxicidad
Stocrin Informacion de Pacientes
SUSTIVA® (sus-TEE-vah) [efavirenz (eh-FAH-vih-rehnz)] capsules and tablets
ALERT: Find out about medicines that should NOT be taken with SUSTIVA.
Please also read the section "MEDICINES YOU SHOULD NOT TAKE WITH SUSTIVA."
Read this information before you start taking SUSTIVA (efavirenz). Read it again each time you refill your prescription, in case there is any new information. This leaflet provides a summary about SUSTIVA and does not include everything there is to know about your medicine. This information is not meant to take the place of talking with your doctor.
What is SUSTIVA?
SUSTIVA is a medicine used in combination with other medicines to help treat infection with Human Immunodeficiency Virus type 1(HIV-1), the virus that causes AIDS (acquired immune deficiency syndrome). SUSTIVA is a type of anti-HIV drug called a "non-nucleoside reverse transcriptase inhibitor" (NNRTI). NNRTIs are not used in the treatment of Human Immunodeficiency Virus type 2 (HIV-2) infection.
SUSTIVA (efavirenz) works by lowering the amount of HIV-1 in the blood (viral load). SUSTIVA must be taken with other anti-HIV medicines. When taken with other anti-HIV medicines, SUSTIVA has been shown to reduce viral load and increase the number of CD4+ cells, a type of immune cell in blood. SUSTIVA may not have these effects in every patient.
SUSTIVA does not cure HIV or AIDS. People taking SUSTIVA may still develop other infections and complications. Therefore, it is very important that you stay under the care of your doctor.
SUSTIVA has not been shown to reduce the risk of passing HIV to others. Therefore, continue to practice safe sex, and do not use or share dirty needles.
What are the possible side effects of SUSTIVA?
Serious psychiatric problems. A small number of patients experience severe depression, strange thoughts, or angry behavior while taking SUSTIVA. Some patients have thoughts of suicide and a few have actually committed suicide. These problems tend to occur more often in patients who have had mental illness. Contact your doctor right away if you think you are having these psychiatric symptoms, so your doctor can decide if you should continue to take SUSTIVA. clinical status. Administration of activated charcoal may Common side effects. Many patients have dizziness, trouble sleeping, drowsiness, trouble concentrating, and/or unusual dreams during treatment with SUSTIVA. These side effects may be reduced if you take SUSTIVA at bedtime on an empty stomach. They also tend to go away after you have taken the medicine for a few weeks. If you have these common side effects, such as dizziness, it does not mean that you will also have serious psychiatric problems, such as severe depression, strange thoughts, or angry behavior. Tell your doctor right away if any of these side effects continue or if they bother you. It is possible that these symptoms may be more severe if SUSTIVA is used with alcohol or mood altering (street) drugs. If you are dizzy, have trouble concentrating, or are drowsy, avoid activities that may be dangerous, such as driving or operating machinery.
Rash is common. Rashes usually go away without any change in treatment. In a small number of patients, rash may be serious. If you develop a rash, call your doctor right away. Rash may be a serious problem in some children. Tell your childís doctor right away if you notice taking SUSTIVA.
Other common side effects include tiredness, upset stomach, vomiting, and diarrhea.
Changes in body fat. Changes in body fat develop in some patients taking anti-HIV medicine. These changes may include an increased amount of fat in the upper back and neck ("buffalo hump"), in the breasts, and around the trunk. Loss of fat from the legs, arms, and face may also happen. The cause and long-term health effects of these fat changes are not known.
Tell your doctor or healthcare provider if you notice any side effects while taking SUSTIVA. Contact your doctor before stopping SUSTIVA because of side effects or for any other reason.
This is not a complete list of side effects possible with SUSTIVA. Ask your doctor or pharmacist for a more complete list of side effects of SUSTIVA and all the medicines you will take.
How should I take SUSTIVA (efavirenz)?
General Information
- You should take SUSTIVA on an empty stomach, preferably at bedtime.
- Swallow SUSTIVA with water.
- Taking SUSTIVA with food increases the amount of medicine in your body, which may increase the frequency of side effects.
- Taking SUSTIVA at bedtime may make some side effects less bothersome.
- SUSTIVA must be taken in combination with other anti-HIV medicines. If you take only SUSTIVA, the medicine may stop working.
- Do not miss a dose of SUSTIVA. If you forget to take SUSTIVA, take the missed dose right away, unless it is almost time for your next dose. Do not double the next dose. Carry on with your regular dosing schedule. If you need help in planning the best times to take your medicine, ask your doctor or pharmacist.
- Take the exact amount of SUSTIVA your doctor prescribes. Never change the dose on your own. Do not stop this medicine unless your doctor tells you to stop.
- If you believe you took more than the prescribed amount of SUSTIVA, contact your local Poison Control Center or emergency room right away.
- Tell your doctor if you start any new medicine or change how you take old ones. Your doses may need adjustment.
- When your SUSTIVA supply starts to run low, get more from your doctor or pharmacy. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time.
The virus may develop resistance to SUSTIVA and become harder to treat.
- Your doctor may want to do blood tests to check for certain side effects while you take SUSTIVA.
Capsules
- The dose of SUSTIVA capsules for adults is 600 mg (three 200-mg capsules, taken together) once a day by mouth. The dose of SUSTIVA for children may be lower.
Tablets
- The dose of SUSTIVA tablets for adults is 600 mg (one tablet) once a day by mouth.
Can children take SUSTIVA?
Yes, children who are able to swallow capsules can take SUSTIVA. Rash may be a serious problem in some children. Tell your childís doctor right away if you SUSTIVA. The dose of SUSTIVA for children may be lower than the dose for adults. Capsules containing lower doses of SUSTIVA are available. Your childís
Who should not take SUSTIVA?
Do not take SUSTIVA if you are allergic to the active ingredient, efavirenz, or to any of the inactive ingredients. Your doctor and pharmacist have a list of the inactive ingredients.
What should I avoid while taking SUSTIVA (efavirenz)?
- Women taking SUSTIVA should not become pregnant. Serious birth defects have been seen in animals treated with SUSTIVA. It is not known whether this could happen in humans. Tell your doctor right away if you are pregnant. Also talk with your doctor if you want to become pregnant.
- Women should not rely only on hormone-based birth control, such as pills, injections, or implants, because SUSTIVA may make these contraceptives ineffective. Women must use a reliable form of barrier contraception, such as a condom or diaphragm, even if they also use other methods of birth control.
- Do not breast-feed if you are taking SUSTIVA. The Centers for Disease Control and Prevention recommend that mothers with HIV not breast-feed because they can pass the HIV through their milk to the baby. Also, SUSTIVA may pass through breast milk and cause serious harm to the baby. Talk with your doctor if you are breast-feeding. You may need to stop breast-feeding or use a different medicine.
- Taking SUSTIVA with alcohol or other medicines causing similar side effects as SUSTIVA, such as drowsiness, may increase those side effects.
- Do not take any other medicines without checking with your doctor. These medicines include prescription and nonprescription medicines and herbal products, especially St.
Before using SUSTIVA, tell your doctor if you
- have problems with your liver or have hepatitis. Your doctor may want to do tests to check your liver while you take SUSTIVA.
- have ever had mental illness or are using drugs or alcohol.
- have ever had seizures or are taking medicine for seizures [for example, Dilantin® (phenytoin), Tegretol® (car-bamazepine), or phenobarbital]. Your doctor may want to check drug levels in your blood from time to time.
What important information should I know about taking other medicines with SUSTIVA?
SUSTIVA may change the effect of other medicines, including ones for HIV, and cause serious side effects.
Your doctor may change your other medicines or change their doses. Other medicines, including herbal products, may affect SUSTIVA. For this reason, it is very important to:
- let all your doctors and pharmacists know that you take SUSTIVA.
- tell your doctors and pharmacists about all medicines you take. This includes those you buy over-the-counter and herbal or natural remedies.
Bring all your prescription and nonprescription medicines as well as any herbal remedies that you are taking when you see a doctor, or make a list of their names, how much you take, and how often you take them. This will give your doctor a complete picture of the medicines you use. Then he or she can decide the best approach for your situation.
Taking SUSTIVA with St. Johnís wort (Hypericum perforatum), an herbal product sold as a dietary supplement, or products containing St. Johnís wort is planning to take St. Johnís wort. Taking St. load and possible resistance to SUSTIVA or cross-resistance to other anti-HIV drugs.
MEDICINES YOU SHOULD NOT TAKE WITH SUSTIVA
The following medicines may cause serious and life-threatening side effects when taken with SUSTIVA. You should not take any of these medicines while taking SUSTIVA:
- Hismanal® (astemizole)
- Propulsid® (cisapride)
- Versed® (midazolam)
- Halcion® (triazolam)
- Ergot medications (for example, Wigraine® and Cafergot® )
The following medicine should not be taken with SUSTIVA since it may lose its effect or may increase the chance of having side effects from SUSTIVA:
- Vfend® (voriconazole)
The following medicines may need to be replaced with another medicine when taken with SUSTIVA (efavirenz):
- Fortovase® , Invirase® (saquinavir)
- Biaxin® (clarithromycin) The following medicines may need to have their dose changed when taken with SUSTIVA:
- Crixivan® (indinavir)
- Kaletra® (lopinavir/ritonavir)
- Methadone
- Mycobutin® (rifabutin)
- Reyataz® (atazanavir). If you are taking SUSTIVA and REYATAZ, you should also be taking Norvir REYATAZ® (ritonavir).
- Zoloft® (sertraline)
These are not all the medicines that may cause problems if you take SUSTIVA. Be sure to tell your doctor about all medicines that you take.
General advice about SUSTIVA:
Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. Do not use SUSTIVA for a condition for which it was not prescribed. Do not give SUSTIVA to other people, even if they have the same symptoms you have. It may harm them.
Keep SUSTIVA at room temperature (77°F) in the bottle can range from 59° to Keep SUSTIVA out of the reach of children.
This leaflet summarizes the most important information about SUSTIVA. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for the full prescribing information about SUSTIVA, or you can visit the SUSTIVA website at http://www.sustiva.com or call 1-800-426-7644.
SUSTIVA® is a registered trademark of Bristol-Myers Squibb Pharma Company. REYATAZ® is a registered trademark of Bristol-Myers Squibb Company. Other brands listed are the trademarks of their respective owners and are not trademarks of Bristol-Myers Squibb Company.