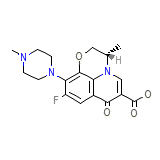
Levaquin Leva-Pak




Categoria
Levaquin Leva-Pak Nombres de marca, Levaquin Leva-Pak Analogos
Levaquin Leva-Pak Marca los nombres de mezcla
Levaquin Leva-Pak Formula quimica
C36H42F2N6O9
Levaquin Leva-Pak RX enlace
http://www.rxlist.com/cgi/generic2/quixin.htm
Levaquin Leva-Pak FDA hoja
Levaquin Leva-Pak MSDS (hoja de seguridad de materiales)
Levaquin Leva-Pak Sintesis de referencia
Mitscher, Lester A. et al;. J.Med.Chem;. 30, 12, 2283 hasta 2286 (1987)
Levaquin Leva-Pak Peso molecular
740.751 g/mol
Levaquin Leva-Pak Punto de fusion
No information avaliable
Levaquin Leva-Pak H2O Solubilidad
Insoluble
Levaquin Leva-Pak Estado
Solid
Levaquin Leva-Pak LogP
1.268
Levaquin Leva-Pak Formas de dosificacion
Tableta, líquido, la solución
Levaquin Leva-Pak Indicacion
Para el tratamiento de la conjuntivitis bacteriana causada por cepas susceptibles de los siguientes organismos: Corynebacterium spp, Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, , Streptococcus (grupos C / F / G), estreptococos del grupo viridans, Acinetobacter lwoffii, Haemophilus influenzae, Serratia marcescens.
Levaquin Leva-Pak Farmacologia
Levofloxacina, una fluoroquinolona antiinfeccioso, es ópticamente activos L-isómero de ofloxacino. Levofloxacino se usa para tratar la conjuntivitis bacteriana, sinusitis, bronquitis crónica, neumonía adquirida en la comunidad y neumonía causada por cepas resistentes a penicilina de Streptococcus pneumoniae, infecciones de la piel y estructura de la piel, infecciones complicadas del tracto urinario y pielonefritis aguda.
Levaquin Leva-Pak Absorcion
La absorción de ofloxacino tras dosis únicas o múltiples de 200 a 400 mg es predecible, y la cantidad de fármaco absorbido aumenta proporcionalmente con la dosis.
Levaquin Leva-Pak Toxicidad
Los efectos secundarios incluyen desorientación, mareos, somnolencia, sofocos y frío, náuseas, dificultad en el habla, la hinchazón y entumecimiento en la cara
Levaquin Leva-Pak Informacion de Pacientes
PATIENT INFORMATION
While taking ofloxacin patient should be advised to:
� to drink fluids liberally;
� that mineral supplements, vitamins with iron or minerals, calcium- , aluminum-, or magnesium-based antacids, sucralfate or Videx�, (Didanosine), chewable/buffered tablets or the pediatric powder for oral solution should not be taken within the two-hour period before or within the two-hour period after taking ofloxacin;
� that ofloxacin can be taken without regard to meals;
� that ofloxacin may cause neurologic adverse effects (e. g. , dizziness, lightheadedness) and that patients should know how they react to ofloxacin before they operate an automobile or machinery or engage in activities requiring mental alertness and coordination;
� to discontinue treatment and inform their physician if they experience pain, inflammation, or rupture of a tendon, and to rest and refrain from exercise until the diagnosis of tendinitis or tendon rupture has been confidently excluded;
� that ofloxacin may be associated with hypersensitivity reactions, even following the first dose, to discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat, difficulty in swallowing or breathing, any swelling suggesting angioedema (e. g. , swelling of the lips, tongue, face; tightness of the throat, hoarseness), or any other symptom of an allergic reaction
� to avoid excessive sunlight or artificial ultraviolet light while receiving ofloxacin and to discontinue therapy if phototoxicity (e. g. , skin eruption) occurs;
� that if they are diabetic and are being treated with insulin or an oral hypoglycemic drug, to discontinue ofloxacin immediately if a hypoglycemic reaction occurs and consult a physician;
� that convulsions have been reported in patients taking quinolones, including ofloxacin, and to notify their physician before taking this drug if there is a history of this condition.
While taking ofloxacin patient should be advised to:
� to drink fluids liberally;
� that mineral supplements, vitamins with iron or minerals, calcium- , aluminum-, or magnesium-based antacids, sucralfate or Videx�, (Didanosine), chewable/buffered tablets or the pediatric powder for oral solution should not be taken within the two-hour period before or within the two-hour period after taking ofloxacin;
� that ofloxacin can be taken without regard to meals;
� that ofloxacin may cause neurologic adverse effects (e. g. , dizziness, lightheadedness) and that patients should know how they react to ofloxacin before they operate an automobile or machinery or engage in activities requiring mental alertness and coordination;
� to discontinue treatment and inform their physician if they experience pain, inflammation, or rupture of a tendon, and to rest and refrain from exercise until the diagnosis of tendinitis or tendon rupture has been confidently excluded;
� that ofloxacin may be associated with hypersensitivity reactions, even following the first dose, to discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat, difficulty in swallowing or breathing, any swelling suggesting angioedema (e. g. , swelling of the lips, tongue, face; tightness of the throat, hoarseness), or any other symptom of an allergic reaction
� to avoid excessive sunlight or artificial ultraviolet light while receiving ofloxacin and to discontinue therapy if phototoxicity (e. g. , skin eruption) occurs;
� that if they are diabetic and are being treated with insulin or an oral hypoglycemic drug, to discontinue ofloxacin immediately if a hypoglycemic reaction occurs and consult a physician;
� that convulsions have been reported in patients taking quinolones, including ofloxacin, and to notify their physician before taking this drug if there is a history of this condition.
Levaquin Leva-Pak Organismos afectados
Bacterias entéricas y otras eubacterias