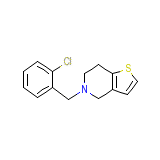
Tazorac




Tazorac Brand names, Tazorac Analogs
Tazorac Brand Names Mixture
- No information avaliable
Tazorac Chemical_Formula
C14H14ClNS
Tazorac RX_link
http://www.rxlist.com/cgi/generic/ticlop.htm
Tazorac fda sheet
Tazorac msds (material safety sheet)
Tazorac Synthesis Reference
No information avaliable
Tazorac Molecular Weight
263.786 g/mol
Tazorac Melting Point
No information avaliable
Tazorac H2O Solubility
Freely soluble
Tazorac State
Solid
Tazorac LogP
3.46
Tazorac Dosage Forms
Tablets (for oral administration, containing 250 mg of ticlopidine hydrochloride)
Tazorac Indication
Used to reduce the risk of thrombotic stroke (fatal or nonfatal) in patients who have experienced stroke precursors, and in patients who have had a completed thrombotic stroke.
Tazorac Pharmacology
Ticlopidine is a platelet aggregation inhibitor structurally and pharmacologically similar to clopidogrel. When taken orally, ticlopidine causes a time- and dose-dependent inhibition of both platelet aggregation and release of platelet granule constituents, as well as a prolongation of bleeding time. The intact drug has no significant in vitro activity at the concentrations attained in vivo; and, although analysis of urine and plasma indicates at least 20 metabolites, no metabolite which accounts for the activity of ticlopidine has been isolated.
Tazorac Absorption
Absorption is greater than 80%. Food increases absorption.
Tazorac side effects and Toxicity
Single oral doses of ticlopidine at 1600 mg/kg and 500 mg/kg were lethal to rats and mice, respectively. Symptoms of acute toxicity were GI hemorrhage, convulsions, hypothermia, dyspnea, loss of equilibrium and abnormal gait.
Tazorac Patient Information
Tazorac Organisms Affected
Humans and other mammals