Tazidime




Tazidime Brand names, Tazidime Analogs
Tazidime Brand Names Mixture
- No information avaliable
Tazidime Chemical_Formula
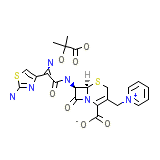
C22H22N6O7S2
Tazidime RX_link
http://www.rxlist.com/cgi/generic2/ceftaz.htm
Tazidime fda sheet
Tazidime msds (material safety sheet)
Tazidime Synthesis Reference
No information avaliable
Tazidime Molecular Weight
546.578 g/mol
Tazidime Melting Point
No information avaliable
Tazidime H2O Solubility
396 mg/L
Tazidime State
Solid
Tazidime LogP
-1.60
Tazidime Dosage Forms
Powder; Powder for solution
Tazidime Indication
For the treatment of patients with infections caused by susceptible strains of organisms in the following diseases: lower respiratory tract infections,skin and skin structure infections, urinary tract infections, bacterial septicemia, bone and joint infections, gynecologic infections, intra abdominal infections (including peritonitis), and central nervous system infections (including meningitis).
Tazidime Pharmacology
Ceftazidime is a semisynthetic, broad-spectrum, beta-lactam antibiotic for parenteral administration. Ceftazidime is bactericidal in action exerting its effect by inhibition of enzymes responsible for cell-wall synthesis. A wide range of gram-negative organisms is susceptible to ceftazidime in vitro, including strains resistant to gentamicin and other aminoglycosides. In addition, ceftazidime has been shown to be active against gram-positive organisms. It is highly stable to most clinically important beta-lactamases, plasmid or chromosomal, which are produced by both gram-negative and gram-positive organisms and, consequently, is active against many strains resistant to ampicillin and other cephalosporins. Ceftazidime has activity against the gram-negative organisms Pseudomonas and Enterobacteriaceae. Its activity against Pseudomonas is a distinguishing feature of ceftazidime among the cephalosporins.
Tazidime Absorption
The absorption of ceftazidime is directly proportional to the size of the dose.
Tazidime side effects and Toxicity
Ceftazidime overdosage has occurred in patients with renal failure. Reactions have included seizure activity, encephalopathy, asterixis, neuromuscular excitability, and coma.
Tazidime Patient Information
No information avaliable
Tazidime Organisms Affected
Enteric bacteria and other eubacteria