Tarka




Tarka Brand names, Tarka Analogs
Tarka Brand Names Mixture
- No information avaliable
Tarka Chemical_Formula
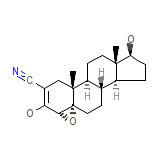
C20H27NO3
Tarka RX_link
No information avaliable
Tarka fda sheet
Tarka msds (material safety sheet)
Tarka Synthesis Reference
No information avaliable
Tarka Molecular Weight
329.433 g/mol
Tarka Melting Point
264 oC
Tarka H2O Solubility
No information avaliable
Tarka State
Solid
Tarka LogP
2.635
Tarka Dosage Forms
Capsules
Tarka Indication
Used in the treatment of Cushing's syndrome. It is normally used in short-term treatment until permanent therapy is possible.
Tarka Pharmacology
Trilostane blocks an enzyme involved in the production of several steroids including cortisol. Inhibiting this enzyme inhibits the production of cortisol. In Cushing's syndrome, the adrenal gland overproduces steroids. Although steroids are important for various functions of the body, too much can cause problems. Trilostane reduces the amount of steroids produced by the adrenal gland. This product was withdrawn from the U.S. market in April 1994.
Tarka Absorption
No information avaliable
Tarka side effects and Toxicity
Symptoms of overdose include darkening of skin, drowsiness or tiredness, loss of appetite, mental depression, skin rash, and/or vomiting.
Tarka Patient Information
No information avaliable
Tarka Organisms Affected
Humans and other mammals