Singular




Singular Brand names, Singular Analogs
Singular Brand Names Mixture
- No information avaliable
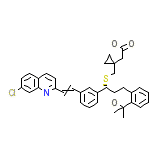
Singular Chemical_Formula
C35H36ClNO3S
Singular RX_link
http://www.rxlist.com/cgi/generic3/monteluk.htm
Singular fda sheet
Singular msds (material safety sheet)
Singular Synthesis Reference
M. L. Belley et al., Eur. Pat. Appl. 480,717 (1992)
Singular Molecular Weight
586.184 g/mol
Singular Melting Point
No information avaliable
Singular H2O Solubility
No information avaliable
Singular State
Solid
Singular LogP
8.488
Singular Dosage Forms
Tablet (oral)
Singular Indication
For the treatment of asthma
Singular Pharmacology
Montelukast, like zafirlukast, is a leukotriene receptor antagonist used as an alternative to anti-inflammatory medications in the management and chronic treatment of asthma and exercise-induced bronchospasm (EIB). Unlike zafirlukast, montelukast does not inhibit CYP2C9 or CYP3A4 and is, therefore, not expected to affect the hepatic clearance of drugs metabolized by these enzymes.
Singular Absorption
Rapidly absorbed following oral administration (bioavailability is 64%)
Singular side effects and Toxicity
Side effects include headache, abdominal or stomach pain, cough, dental pain, dizziness, fever, heartburn, skin rash, stuffy nose, weakness or unusual tiredness.
Singular Patient Information
General
- Patients should be advised to take montelukast daily as prescribed, even when they are asymptomatic, as well as during periods of worsening asthma, and to contact their physicians if their asthma is not well controlled.
- Patients should be advised that oral tablets of montelukast are not for the treatment of acute asthma attacks. They should have appropriate short-acting inhaled b-agonist medication available to treat asthma exacerbations.
- Patients should be advised that, while using montelukast, medical attention should be sought if short-acting inhaled bronchodilators are needed more often than usual, or if more than the maximum number of inhalations of short-acting bronchodilator treatment prescribed for 24-hour period are needed.
- Patients receiving montelukast should be instructed not to decrease the dose or stop taking any other antiasthma medications unless instructed by a physician.
- Patients who have exacerbations of asthma after exercise should be instructed to continue to use their usual regimen of inhaled b-agonists as prophylaxis unless otherwise instructed by their physician. All patients should have available for rescue a short-acting inhaled b-agonist.
- Patients with known aspirin sensitivity should be advised to continue avoidance of aspirin or non-steroidal anti-inflammatory agents while taking montelukast.
Phenylketonurics: Phenylketonuric patients should be informed that the chewable tablet contains phenylalanine (a component of aspartame) 0.842 mg per 5-mg chewable tablet.
Singular Organisms Affected
Humans and other mammals