Sabril (TN)




Sabril (TN) Brand names, Sabril (TN) Analogs
Sabril (TN) Brand Names Mixture
- No information avaliable
Sabril (TN) Chemical_Formula
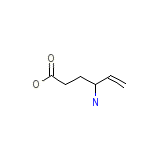
C6H11NO2
Sabril (TN) RX_link
No information avaliable
Sabril (TN) fda sheet
Sabril (TN) msds (material safety sheet)
Sabril (TN) Synthesis Reference
No information avaliable
Sabril (TN) Molecular Weight
129.157 g/mol
Sabril (TN) Melting Point
No information avaliable
Sabril (TN) H2O Solubility
55.1 mg/mL
Sabril (TN) State
Solid
Sabril (TN) LogP
0.056
Sabril (TN) Dosage Forms
Powder (500 mg sachets); Tablet (500 mg)
Sabril (TN) Indication
For use as an adjunctive treatment (with other drugs) in treatment resistant epilepsy, complex partial seizures, secondary generalized seizures, and for monotherapy use in infantile spasms in West syndrome.
Sabril (TN) Pharmacology
Vigabatrin, is an anticonvulsant chemically unrelated to other anticonvulsants. Vigabatrin inhibits the catabolism of GABA. It is an analog of GABA, but it is not a receptor agonist.
Sabril (TN) Absorption
Rapidly absorbed following oral administration. Food may slightly decrease the rate, but not the extent, of absorption.
Sabril (TN) side effects and Toxicity
No information avaliable
Sabril (TN) Patient Information
This medicine may reduce your ability to drive or operate machinery safely. Do not drive or operate machinery until you know how this medicine affects you and you are sure it won't affect your performance. Avoid stopping this medicine suddenly. The dose of this medicine should be gradually decreased over a period of two to four weeks. After months to years of taking this medication, damage to vision may occur. Regular eye examinations should be conducted and notify your doctor if any change in vision occurs.
Sabril (TN) Organisms Affected
Humans and other mammals