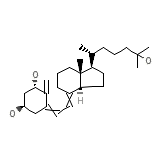
Rocaltrol (TN)




Rocaltrol (TN) Brand names, Rocaltrol (TN) Analogs
Rocaltrol (TN) Brand Names Mixture
- No information avaliable
Rocaltrol (TN) Chemical_Formula
C27H44O3
Rocaltrol (TN) RX_link
http://www.rxlist.com/cgi/generic2/calcitri.htm
Rocaltrol (TN) fda sheet
Rocaltrol (TN) msds (material safety sheet)
Rocaltrol (TN) Synthesis Reference
No information avaliable
Rocaltrol (TN) Molecular Weight
416.636 g/mol
Rocaltrol (TN) Melting Point
113 oC
Rocaltrol (TN) H2O Solubility
Insoluble
Rocaltrol (TN) State
Solid
Rocaltrol (TN) LogP
4.96
Rocaltrol (TN) Dosage Forms
Capsules; Solution
Rocaltrol (TN) Indication
Used to treat vitamin D deficiency or insufficiency, refractory rickets (vitamin D resistant rickets), familial hypophosphatemia and hypoparathyroidism, and in the management of hypocalcemia and renal osteodystrophy in patients with chronic renal failure undergoing dialysis. Also used in conjunction with calcium in the management and prevention of primary or corticosteroid-induced osteoporosis.
Rocaltrol (TN) Pharmacology
Calcitriol, a pharmaceutical form of vitamin D, has anti-osteoporotic, immunomodulatory, anticarcinogenic, antipsoriatic, antioxidant, and mood-modulatory activities. Calcitriol has been found to be effective in the treatment of psoriasis when applied topically. Calcitriol has been found to induce differentiation and/or inhibit cell proliferation in a number of malignant cell lines including human prostate cancer cells. Vitamin D deficiency has long been suspected to increase the susceptibility to tuberculosis. The active form of Calcitriol, 1,25 (OH)2 D, has been found to enhance the ability of mononuclear phagocytes to suppress the intracellular growth of Mycobacterium tuberculosis. 1,25(OH)2D has demonstrated beneficial effects in animal models of such autoimmune diseases as rheumatoid arthritis. It has also been found to induce monocyte differentiation and to inhibit lymphocyte proliferation and production of cytokines, including interleukin IL-1 and IL-2, as well as to suppress immunoglobulin secretion by B lymphocytes. Vitamin D appears to demonstrate both immune-enhancing and immunosuppressive effects.
Rocaltrol (TN) Absorption
Rapidly absorbed from the intestine.
Rocaltrol (TN) side effects and Toxicity
LD50 (oral, rat) = 620 μg/kg; LD50 (intraperitoneal, rat) > 5 mg/kg; Overdose evident in elevated blood calcium levels causing symptoms of anorexia, nausea and vomiting, polyuria, polydipsia, weakness, pruritus, and nervousness, potentially with irreversible calcification of soft tissue in the kidney and liver.
Rocaltrol (TN) Patient Information
Patient information for vitamin D analogues (Vitamin D2, Vitamin D3, Calcitriol, and Calcidiol):
The patient and his or her parents or s.o.s. should be informed about compliance with dosage instructions, adherence to instructions about diet and calcium supplementation and avoidance of the use of unapproved nonprescription drugs. Patients should also be carefully informed about the symptoms of hypercalcemia.
The effectiveness of vitamin D therapy is predicated on the assumption that each patient is receiving an adequate daily intake of calcium. Patients are advised to have a dietary intake of calcium at a minimum of 600 mg daily. The U.S. RDA for calcium in adults is 800 mg to 1200 mg.
The patient and his or her parents or s.o.s. should be informed about compliance with dosage instructions, adherence to instructions about diet and calcium supplementation and avoidance of the use of unapproved nonprescription drugs. Patients should also be carefully informed about the symptoms of hypercalcemia.
The effectiveness of vitamin D therapy is predicated on the assumption that each patient is receiving an adequate daily intake of calcium. Patients are advised to have a dietary intake of calcium at a minimum of 600 mg daily. The U.S. RDA for calcium in adults is 800 mg to 1200 mg.
Rocaltrol (TN) Organisms Affected
Humans and other mammals