Miglustat, Hydrochloride




Miglustat, Hydrochloride Brand names, Miglustat, Hydrochloride Analogs
Miglustat, Hydrochloride Brand Names Mixture
- No information avaliable
Miglustat, Hydrochloride Chemical_Formula
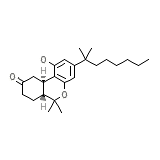
C24H36O3
Miglustat, Hydrochloride RX_link
No information avaliable
Miglustat, Hydrochloride fda sheet
Miglustat, Hydrochloride msds (material safety sheet)
Miglustat, Hydrochloride Synthesis Reference
No information avaliable
Miglustat, Hydrochloride Molecular Weight
372.541 g/mol
Miglustat, Hydrochloride Melting Point
No information avaliable
Miglustat, Hydrochloride H2O Solubility
No information avaliable
Miglustat, Hydrochloride State
Solid
Miglustat, Hydrochloride LogP
6.909
Miglustat, Hydrochloride Dosage Forms
Capsules (0.1 mg)
Miglustat, Hydrochloride Indication
Used for the control of nausea and vomiting, caused by chemotherapeutic agents used in the treatment of cancer, in patients who have failed to respond adequately to conventional antiemetic treatments.
Miglustat, Hydrochloride Pharmacology
Nabilone is a cannabinoid with therapeutic uses. It is an analog of dronabinol (also known as tetrahydrocannabinol or THC), the psychoactive ingredient in cannabis. It is reserved for use in individuals who do not respond to the more commonly used anti-emetics. This is mainly because cannabinoids have potential adverse effects similar to that of cannabis and may cause changes in mood and behaviour.
Miglustat, Hydrochloride Absorption
Rapidly absorbed from the gastrointestinal tract following oral administration.
Miglustat, Hydrochloride side effects and Toxicity
Symptoms of overdose include difficulty in breathing, hallucinations, mental changes (severe), nervousness or anxiety (severe). Monkeys treated with Nabilone at doses as high as 2mg/kg/day for a year experienced no significant adverse events. This result contrasts with the finding in a planned 1-year dog study that was prematurely terminated because of deaths associated with convulsions in dogs receiving as little as 0.5mg/kg/day. The earliest deaths, however, occurred at 56 days in dogs receiving 2mg/kg/day. The unusual vulnerability of the dog is not understood; it is hypothesised, however, that the explanation lies in the fact that the dog differs markedly from other species (including humans) in its metabolism of Nabilone.
Miglustat, Hydrochloride Patient Information
Miglustat, Hydrochloride Organisms Affected
Humans and other mammals