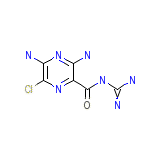
Midamor




Midamor Brand names, Midamor Analogs
Midamor Brand Names Mixture
- Blu Kote Dr Naylors (Acriflavine + Gentian Violet + Glycerine + Isopropyl Alcohol + Sodium Propionate + Urea)
- Blue Lotion (Gentian Violet + Methylene Blue + Tannic Acid)
- Co-Op Pinkeye Spray (Gentian Violet + Neomycin (Neomycin Sulfate))
- Cristisol (Acriflavine + Gentian Violet + Isopropyl Alcohol)
- Pink Eye Guard (Allantoin + Boric Acid + Gentian Violet)
- Pink Eye Spray (Furfural + Gentian Violet + Isopropyl Alcohol + Methylene Blue + Phenylmercuric Nitrate (Basic) + Tetrahydrofurfuryl Alcohol + Urea)
- Pinkeye Spray (Gentian Violet + Neomycin (Neomycin Sulfate))
- Triple Dye (Brilliant Green + Gentian Violet + Proflavine Hemisulfate)
- Wound & Pinkeye Spray (Gentian Violet + Neomycin (Neomycin Sulfate))
- Wound Kote Gentian Violet Spray Wound Dre (Acriflavine + Furfural + Methyl Violet + Sodium Propionate + Urea)
Midamor Chemical_Formula
C6H8ClN7O
Midamor RX_link
http://www.rxlist.com/cgi/generic2/amilor.htm
Midamor fda sheet
Midamor msds (material safety sheet)
Midamor Synthesis Reference
Cragoe, Belg, pat. 639,386 (1964 to Merck & Co.), C.A. 62, 14698f (1965)
Midamor Molecular Weight
229.627 g/mol
Midamor Melting Point
240 oC
Midamor H2O Solubility
Slightly soluble
Midamor State
Solid
Midamor LogP
-1.245
Midamor Dosage Forms
Tablets (oral, 5mg)
Midamor Indication
For use as adjunctive treatment with thiazide diuretics or other kaliuretic-diuretic agents in congestive heart failure or hypertension.
Midamor Pharmacology
Amiloride, an antikaliuretic-diuretic agent, is a pyrazine-carbonyl-guanidine that is unrelated chemically to other known antikaliuretic or diuretic agents. It is an antihypertensive, potassium-sparing diuretic that was first approved for use in 1967 and helps to treat hypertension and congestive heart failure. The drug is often used in conjunction with thiazide or loop diuretics. Due to its potassium-sparing capacities, hyperkalemia (high blood potassium levels) are occasionally observed in patients taking amiloride. The risk is high in concurrent use of ACE inhibitors or spironolactone. Patients are also advised not to use potassium-containing salt replacements.
Midamor Absorption
Readily absorbed following oral administration.
Midamor side effects and Toxicity
No data are available in regard to overdosage in humans. The oral LD50 of amiloride hydrochloride (calculated as the base) is 56 mg/kg in mice and 36 to 85 mg/kg in rats, depending on the strain. The most likely signs and symptoms to be expected with overdosage are dehydration and electrolyte imbalance.
Midamor Patient Information
No information avaliable
Midamor Organisms Affected
Humans and other mammals