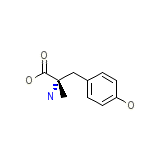
Methylcyclothiazide




Methylcyclothiazide Brand names, Methylcyclothiazide Analogs
Methylcyclothiazide Brand Names Mixture
- No information avaliable
Methylcyclothiazide Chemical_Formula
C10H13NO3
Methylcyclothiazide RX_link
http://www.rxlist.com/cgi/generic2/metyrosine.htm
Methylcyclothiazide fda sheet
Methylcyclothiazide msds (material safety sheet)
Methylcyclothiazide Synthesis Reference
No information avaliable
Methylcyclothiazide Molecular Weight
195.215 g/mol
Methylcyclothiazide Melting Point
312.5 oC
Methylcyclothiazide H2O Solubility
Very slightly soluble
Methylcyclothiazide State
Solid
Methylcyclothiazide LogP
-1.475
Methylcyclothiazide Dosage Forms
Capsules for oral administration (250 mg)
Methylcyclothiazide Indication
For use in the treatment of patients with pheochromocytoma for preoperative preparation of patients for surgery, management of patients when surgery is contraindicated, and chronic treatment of patients with malignant pheochromocytoma.
Methylcyclothiazide Pharmacology
In patients with pheochromocytoma, who produce excessive amounts of norepinephrine and epinephrine, administration of one to four grams of metyrosine per day has reduced catecholamine biosynthesis from about 35 to 80 percent as measured by the total excretion of catecholamines and their metabolites (metanephrine and vanillylmandelic acid). The maximum biochemical effect usually occurs within two to three days, and the urinary concentration of catecholamines and their metabolites usually returns to pretreatment levels within three to four days after metyrosine is discontinued. Most patients with pheochromocytoma treated with metyrosine experience decreased frequency and severity of hypertensive attacks with their associated headache, nausea, sweating, and tachycardia. In patients who respond, blood pressure decreases progressively during the first two days of therapy with metyrosine; after withdrawal, blood pressure usually increases gradually to pretreatment values within two to three days.
Methylcyclothiazide Absorption
Well absorbed from the gastrointestinal tract.
Methylcyclothiazide side effects and Toxicity
Signs of metyrosine overdosage include those central nervous system effects observed in some patients even at low dosages. At doses exceeding 2000 mg/day, some degree of sedation or feeling of fatigue may persist. Doses of 2000-4000 mg/day can result in anxiety or agitated depression, neuromuscular effects (including fine tremor of the hands, gross tremor of the trunk, tightening of the jaw with trismus), diarrhea, and decreased salivation with dry mouth. The acute toxicity of metyrosine was 442 mg/kg and 752 mg/kg in the female mouse and rat respectively.
Methylcyclothiazide Patient Information
No information avaliable
Methylcyclothiazide Organisms Affected
Humans and other mammals