Meclofenamate




Meclofenamate Brand names, Meclofenamate Analogs
Meclofenamate Brand Names Mixture
- No information avaliable
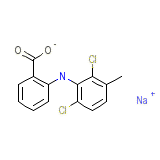
Meclofenamate Chemical_Formula
C14H11Cl2NO2
Meclofenamate RX_link
http://www.rxlist.com/cgi/generic3/meclofen.htm
Meclofenamate fda sheet
Meclofenamate msds (material safety sheet)
Meclofenamate Synthesis Reference
No information avaliable
Meclofenamate Molecular Weight
295.02 g/mol
Meclofenamate Melting Point
257 oC
Meclofenamate H2O Solubility
30 mg/L
Meclofenamate State
Solid
Meclofenamate LogP
4.998
Meclofenamate Dosage Forms
Capsules (containing 50 mg or 100 mg meclofenamic acid as the sodium salt)
Meclofenamate Indication
For the relief of mild to moderate pain, for the treatment of primary dysmenorrhea and for the treatment of idiopathic heavy menstrual blood loss. Also for relief of the signs and symptoms of acute and chronic rheumatoid arthritis and osteoarthritis.
Meclofenamate Pharmacology
Meclofenamate is a nonsteroidal agent which has demonstrated anti-inflammatory, analgesic, and antipyretic activity in laboratory animals.
Meclofenamate Absorption
Rapidly absorbed in man following single and multiple oral doses with peak plasma concentrations occurring in 0.5 to 2 hours. The concomitant administration of antacids (aluminum and magnesium hydroxides) does not interfere with absorption of meclofenamate. Unlike most NSAIDs, which when administered with food have a decrease in rate but not in extent of absorption, meclofenamic acid is decreased in both. It has been reported that following the administration of meclofenamate capsules one-half hour after a meal, the average extent of bioavailability decreased by 26%, the average peak concentration (Cmax) decreased fourfold and the time to Cmax was delayed by 3 hours.
Meclofenamate side effects and Toxicity
No information avaliable
Meclofenamate Patient Information
Meclofenamate Organisms Affected
Humans and other mammals