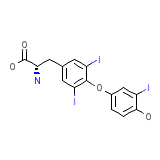
LEVITIRACETAM




LEVITIRACETAM Brand names, LEVITIRACETAM Analogs
LEVITIRACETAM Brand Names Mixture
- No information avaliable
LEVITIRACETAM Chemical_Formula
C15H12I3NO4
LEVITIRACETAM RX_link
http://www.rxlist.com/cgi/generic3/liothyronine.htm
LEVITIRACETAM fda sheet
LEVITIRACETAM msds (material safety sheet)
LEVITIRACETAM Synthesis Reference
No information avaliable
LEVITIRACETAM Molecular Weight
650.974 g/mol
LEVITIRACETAM Melting Point
236.5 oC
LEVITIRACETAM H2O Solubility
3.96 mg/L
LEVITIRACETAM State
Solid
LEVITIRACETAM LogP
3.001
LEVITIRACETAM Dosage Forms
Tablet
LEVITIRACETAM Indication
Used as replacement or supplemental therapy in patients with hypothyroidism of any etiology, except transient hypothyrodism during the recovery phase of subacute thyroiditis.
LEVITIRACETAM Pharmacology
Thyroid hormone drugs are natural or synthetic preparations containing tetraiodothyronine (T4, levothyroxine) or triiodothyronine (T3, liothyronine) or both. T4 and T3 are produced in the human thyroid gland by the iodination and coupling of the amino acid tyrosine. Liothyronine (T3) contains three atoms of iodine and is formed by the coupling of one molecule of diiodotyrosine (DIT) with one molecule of monoiodotyrosine (MIT). These hormones enhance oxygen consumption by most tissues of the body and increase the basal metabolic rate and the metabolism of carbohydrates, lipids and proteins. Thus, they exert a profound influence on every organ system in the body and are of particular importance in the development of the central nervous system.
LEVITIRACETAM Absorption
Almost totally absorbed, 95 percent in 4 hours.
LEVITIRACETAM side effects and Toxicity
No information avaliable
LEVITIRACETAM Patient Information
LEVITIRACETAM Organisms Affected
Humans and other mammals