Glyset




Glyset Brand names, Glyset Analogs
Glyset Brand Names Mixture
- No information avaliable
Glyset Chemical_Formula
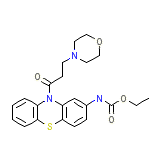
C22H25N3O4S
Glyset RX_link
No information avaliable
Glyset fda sheet
Glyset msds (material safety sheet)
Glyset Synthesis Reference
No information avaliable
Glyset Molecular Weight
427.518 g/mol
Glyset Melting Point
156-157 oC
Glyset H2O Solubility
0.457 mg/L
Glyset State
Solid
Glyset LogP
3.207
Glyset Dosage Forms
Film-coated tablets (200-mg, 250-mg, or 300-mg)
Glyset Indication
Used to treat irregular heartbeats (arrhythmias) and maintain a normal heart rate.
Glyset Pharmacology
Moricizine is used to treat irregular heartbeats (arrhythmias) and to maintain a normal heart rate. It acts on the heart muscle to improve the heart's rhythm. Moricizine has potent local anesthetic activity and membrane stabilizing effect. Decreases excitability, conduction velocity, and automaticity as a result of slowed atrioventricular (AV) nodal and His-Purkinje conduction. Decreases the action potential duration (APD) in Purkinje fibers; also decreases the effective refractory period (ERP) but to a lesser extent than the APD, so the ERP/APD ratio is increased. Decreases the maxiumum rate of Phase 0 depolarization (V max ), but does not affect action potential amplitude or maximum diastolic potential. Does not affect atrial, AV nodal, or left ventricular refractory periods and has minimal effect on ventricular repolarization (evidenced by the overall decrease in JT interval). Has no effect on sinoatrial (SA) nodal or intra-atrial conduction and only minimal effect on sinus cycle length and sinus node recovery time. In the Vaughan Williams classification of antiarrhythmics, moricizine is considered to be a class I agent. It has properties of class IA, IB, and IC agents but does not clearly belong to any of the three subclasses. It has less effect on the slope of phase 0 and a greater effect on action potential duration and effective refractory period than class IC agents.
Glyset Absorption
Well absorbed, absorption is complete within 2 to 3 hours. Significant first-pass metabolism results in an absolute bioavailability of approximately 38%. Administration within 30 minutes after a meal slows the rate, but does not affect the extent of absorption, although peak plasma concentrations are reduced.
Glyset side effects and Toxicity
Symptoms of overdose include vomiting, unconsciousness, and severe low blood pressure.
Glyset Patient Information
Glyset Organisms Affected
Humans and other mammals