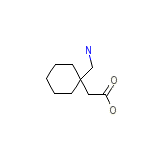
Gabapentine




Gabapentine Brand names, Gabapentine Analogs
Gabapentine Brand Names Mixture
- No information avaliable
Gabapentine Chemical_Formula
C9H17NO2
Gabapentine RX_link
http://www.rxlist.com/cgi/generic/gabapent.htm
Gabapentine fda sheet
Gabapentine msds (material safety sheet)
Gabapentine Synthesis Reference
G Satzinger et al., U.S. Pat. 4,024,175 (1977).
Gabapentine Molecular Weight
171.237 g/mol
Gabapentine Melting Point
162-166 oC
Gabapentine H2O Solubility
4490 mg/L
Gabapentine State
Solid
Gabapentine LogP
0.828
Gabapentine Dosage Forms
Capsule; Tablet (oral); Solution (oral)
Gabapentine Indication
For the management of postherpetic neuralgia in adults and as adjunctive therapy in the treatment of partial seizures with and without secondary generalization in patients over 12 years of age with epilepsy.
Gabapentine Pharmacology
Gabapentin, an analog of GABA, is used as an anticonvulsant to treat partial seizures, amyotrophic lateral sclerosis (ALS), and painful neuropathies. Potential uses include monotherapy of refractory partial seizure disorders, and treatment of spasticity in multiple sclerosis, tremor. mood disorders, and attenuation of disruptive behaviors in dementia. Gabapentin has high lipid solubility, is not metabolized by the liver, has no protein binding, and doesn't possess the usual drug interactions.
Gabapentine Absorption
Rapid. Absorbed in part by the L-amino acid transport system, which is a carrier-mediated, saturable transport system; as the dose increases, bioavailability decreases. Bioavailability ranges from approximately 60% for a 900 mg dose per day to approximately 27% for a 4800 milligram dose per day. Food has a slight effect on the rate and extent of absorption of gabapentin (14% increase in AUC).
Gabapentine side effects and Toxicity
Symptoms of overdose include ataxia, labored breathing, ptosis, sedation, hypoactivity, and excitation.
Gabapentine Patient Information
Patients should be instructed to take Neurontin only as prescribed.
Patients should be advised that Neurontin may cause dizziness, somnolence and other symptoms and signs of CNS depression. Accordingly, they should be advised neither to drive a car nor to operate other complex machinery until they have gained sufficient experience on Neurontin to gauge whether or not it affects their mental and/or motor performance adversely. Patients who require concomitant treatment with morphine may experience increases in gabapentin concentrations. Patients should be carefully observed for signs of CNS depression, such as somnolence, and the dose of Neurontin or morphine should be reduced appropriately.
Gabapentine Organisms Affected
Humans and other mammals