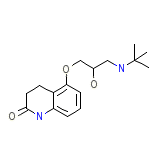
Endak Hydrochloride




Endak Hydrochloride Brand names, Endak Hydrochloride Analogs
Endak Hydrochloride Brand Names Mixture
- No information avaliable
Endak Hydrochloride Chemical_Formula
C16H24N2O3
Endak Hydrochloride RX_link
http://www.rxlist.com/cgi/generic2/carteol.htm
Endak Hydrochloride fda sheet
Endak Hydrochloride msds (material safety sheet)
Endak Hydrochloride Synthesis Reference
No information avaliable
Endak Hydrochloride Molecular Weight
292.373 g/mol
Endak Hydrochloride Melting Point
No information avaliable
Endak Hydrochloride H2O Solubility
No information avaliable
Endak Hydrochloride State
Solid
Endak Hydrochloride LogP
2.037
Endak Hydrochloride Dosage Forms
Ophthalmic Solution (1%)
Endak Hydrochloride Indication
For the treatment of intraocular hypertension and chronic open-angle glaucoma
Endak Hydrochloride Pharmacology
Carteolol is a beta1 and beta2 (non-selective) adrenergic receptor-blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity. Carteolol, when applied topically to the eye, has the action of reducing elevated, as well as normal, intraocular pressure, whether or not accompanied by glaucoma. Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss and optic nerve damage. Carteolol reduces intraocular pressure with little or no effect on pupil size or accommodation in contrast to the miosis which cholinergic agents are known to produce.
Endak Hydrochloride Absorption
No information avaliable
Endak Hydrochloride side effects and Toxicity
The most common effects expected with overdosage of a beta-adrenergic blocking agent are bradycardia, bronchospasm, congestive heart failure and hypotension.
Endak Hydrochloride Patient Information
No information avaliable
Endak Hydrochloride Organisms Affected
Humans and other mammals