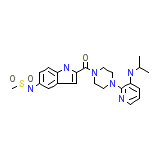
DLV




DLV Brand names, DLV Analogs
DLV Brand Names Mixture
- No information avaliable
DLV Chemical_Formula
C22H28N6O3S
DLV RX_link
No information avaliable
DLV fda sheet
DLV msds (material safety sheet)
DLV Synthesis Reference
D. L. Romero et al., PCT Int. pat. Appl. 91 09,849 (1991)
DLV Molecular Weight
456.562 g/mol
DLV Melting Point
226-228 oC
DLV H2O Solubility
No information avaliable
DLV State
Solid
DLV LogP
2.926
DLV Dosage Forms
Caplet
DLV Indication
For the treatment of HIV-1 infection in combination with appropriate antiretroviral agents when therapy is warranted
DLV Pharmacology
Delavirdine is a non-nucleoside reverse transcriptase inhibitor (nNRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1). Delavirdine binds directly to reverse transcriptase (RT) and blocks the RNA-dependent and DNA-dependent DNA polymerase activities by causing a disruption of the enzyme's catalytic site. The activity of Delavirdine does not compete with template or nucleoside triphosphates. HIV-2 RT and eukaryotic DNA polymerases (such as human DNA polymerases alpha, beta, or sigma) are not inhibited by Delavirdine.
DLV Absorption
Rapidly absorbed
DLV side effects and Toxicity
Major toxicity of delavirdine is rash and should be advised to promptly notify their physician should rash occur. The majority of rashes associated with delavirdine occur within 1 to 3 weeks after initiating treatment with delavirdine. The rash normally resolves in 3 to 14 days and may be treated symptomatically while therapy with delavirdine is continued. Any patient experiencing severe rash or rash accompanied by symptoms such as fever, blistering, oral lesions, conjunctivitis, swelling, muscle or joint aches should discontinue medication and consult a physician.
DLV Patient Information
Patients should be informed that RESCRIPTOR is not a cure for HIV-1
infection and that they may continue to acquire illnesses associated
with HIV-1 infection, including opportunistic infections. Treatment
with RESCRIPTOR has not been shown to reduce the incidence or frequency
of such illnesses, and patients should be advised to remain under the
care of a physician when using RESCRIPTOR. Patients should be advised
that the long-term effects of treatment with RESCRIPTOR are unknown at
this time. They should be advised that the use of RESCRIPTOR has not been
shown to reduce the risk of transmission of HIV-1.
infection and that they may continue to acquire illnesses associated
with HIV-1 infection, including opportunistic infections. Treatment
with RESCRIPTOR has not been shown to reduce the incidence or frequency
of such illnesses, and patients should be advised to remain under the
care of a physician when using RESCRIPTOR. Patients should be advised
that the long-term effects of treatment with RESCRIPTOR are unknown at
this time. They should be advised that the use of RESCRIPTOR has not been
shown to reduce the risk of transmission of HIV-1.
DLV Organisms Affected
Human immunodeficiency virus