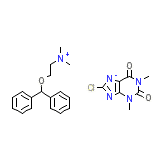
Cedilanid-d




Cedilanid-d Brand names, Cedilanid-d Analogs
Cedilanid-d Brand Names Mixture
- No information avaliable
Cedilanid-d Chemical_Formula
C24H28ClN5O3
Cedilanid-d RX_link
No information avaliable
Cedilanid-d fda sheet
Cedilanid-d msds (material safety sheet)
Cedilanid-d Synthesis Reference
No information avaliable
Cedilanid-d Molecular Weight
469.964 g/mol
Cedilanid-d Melting Point
204.5 oC
Cedilanid-d H2O Solubility
3000 mg/L
Cedilanid-d State
Solid
Cedilanid-d LogP
-0.39
Cedilanid-d Dosage Forms
Capsule (sustained-release); Liquid; Solution; Suppository; Syrup; Tablet; Tablet (extended-release)
Cedilanid-d Indication
Used for treating vertigo, motion sickness, and nausea associated with pregnancy.
Cedilanid-d Pharmacology
Dimenhydrinate is an antiemetics drug combination that contains diphenhydramine and theophylline. It is not effective in the treatment of nausea associated with cancer chemotherapy. Dimenhydrinate directly inhibits the stimulation of certain nerves in the brain and inner ear to suppress nausea, vomiting, dizziness, and vertigo.
Cedilanid-d Absorption
Well absorbed after oral administration.
Cedilanid-d side effects and Toxicity
Symptoms of overdose include delerium and hallucinations.
Cedilanid-d Patient Information
No information avaliable
Cedilanid-d Organisms Affected
Humans and other mammals