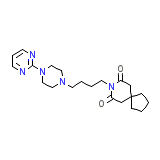
Buspirone HCL




Buspirone HCL Brand names, Buspirone HCL Analogs
Buspirone HCL Brand Names Mixture
- No information avaliable
Buspirone HCL Chemical_Formula
C21H31N5O2
Buspirone HCL RX_link
http://www.rxlist.com/cgi/generic/buspir.htm
Buspirone HCL fda sheet
Buspirone HCL msds (material safety sheet)
Buspirone HCL Synthesis Reference
U.S. Pat. 3,717,634 (1973)
Buspirone HCL Molecular Weight
385.503 g/mol
Buspirone HCL Melting Point
201.5-202.5oC
Buspirone HCL H2O Solubility
21.4 mg/L
Buspirone HCL State
Solid
Buspirone HCL LogP
1.238
Buspirone HCL Dosage Forms
Tablet (5 mg, 10 mg)
Buspirone HCL Indication
For the management of anxiety disorders or the short-term relief of the symptoms of anxiety, and also as an augmention of SSRI-treatment against depression.
Buspirone HCL Pharmacology
Buspirone is used in the treatment of generalized anxiety where it has advantages over other antianxiety drugs because it does not cause sedation (drowsiness) and does not cause tolerance or physical dependence. Buspirone differs from typical benzodiazepine anxiolytics in that it does not exert anticonvulsant or muscle relaxant effects. It also lacks the prominent sedative effect that is associated with more typical anxiolytics. in vitro preclinical studies have shown that buspirone has a high affinity for serotonin (5-HT1A) receptors. Buspirone has no significant affinity for benzodiazepine receptors and does not affect GABA binding in vitro or in vivo when tested in preclinical models. Buspirone has moderate affinity for brain D2-dopamine receptors. Some studies do suggest that buspirone may have indirect effects on other neurotransmitter systems.
Buspirone HCL Absorption
Rapidly absorbed in man. Bioavailability is low and variable (approximately 5%) due to extensive first pass metabolism.
Buspirone HCL side effects and Toxicity
Oral, rat LD50 = 136 mg/kg. Symptoms of overdose include dizziness, drowsiness, nausea or vomiting, severe stomach upset, and unusually small pupils.
Buspirone HCL Patient Information
Buspirone is used for the treatment of nervousness and anxiety. Optimum results are usually seen after three to four weeks of treatment. This medication may be taken with or without food. Inform your physican if you are pregnant or nursing. Buspirone may cause dizziness and drowsiness; use caution while driving or operating hazardous machinery. Do not take any other sedating drugs or drink alcohol while taking this medication. Do not take this medication with a monoamine oxidase inhibitor. Notify physician if you develop muscle spasms, uncontrolled twitching in the face and body, or uncontrolled tongue movements.
Buspirone HCL Organisms Affected
Humans and other mammals