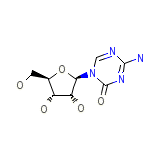
Azacytidine




Azacytidine Brand names, Azacytidine Analogs
Azacytidine Brand Names Mixture
- No information avaliable
Azacytidine Chemical_Formula
C8H12N4O5
Azacytidine RX_link
http://www.rxlist.com/cgi/generic3/vidaza.htm
Azacytidine fda sheet
Azacytidine msds (material safety sheet)
Azacytidine Synthesis Reference
No information avaliable
Azacytidine Molecular Weight
244.205 g/mol
Azacytidine Melting Point
229 oC
Azacytidine H2O Solubility
8.9E+004 mg/L
Azacytidine State
Solid
Azacytidine LogP
-3.017
Azacytidine Dosage Forms
Azacitidine for injectable suspension is supplied as a lyophilized powder in 100 mg single-use vials packaged in cartons of 1 vial.
Azacytidine Indication
For treatment of patients with the following myelodysplastic syndrome subtypes: refractory anemia or refractory anemia with ringed sideroblasts (if accompanied by neutropenia or thrombocytopenia or requiring transfusions), refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
Azacytidine Pharmacology
Azacitidine is believed to exert its antineoplastic effects by causing hypomethylation of DNA and direct cytotoxicity on abnormal hematopoietic cells in the bone marrow. The concentration of azacitidine required for maximum inhibition of DNA methylation in vitro does not cause major suppression of DNA synthesis. Hypomethylation may restore normal function to genes that are critical for differentiation and proliferation. The cytotoxic effects of azacitidine cause the death of rapidly dividing cells, including cancer cells that are no longer responsive to normal growth control mechanisms. Non-proliferating cells are relatively insensitive to azacitidine.
Azacytidine Absorption
Azacitidine is rapidly absorbed after subcutaneous administration. The bioavailability of subcutaneous azacitidine relative to IV azacitidine is approximately 89%, based on area under the curve.
Azacytidine side effects and Toxicity
One case of overdose with azacitidine was reported during clinical trials. A patient experienced diarrhea, nausea, and vomiting after receiving a single IV dose of approximately 290 mg/m2, almost 4 times the recommended starting dose.
Azacytidine Patient Information
Azacytidine Organisms Affected
Humans and other mammals