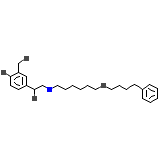
Arial




Arial Brand names, Arial Analogs
Arial Brand Names Mixture
- Advair 100 Diskus (Fluticasone Propionate + Salmeterol Xinafoate)
- Advair 125 (Fluticasone Propionate + Salmeterol Xinafoate)
- Advair 250 (Fluticasone Propionate + Salmeterol Xinafoate)
- Advair 250 Diskus (Fluticasone Propionate + Salmeterol Xinafoate)
- Advair 500 Diskus (Fluticasone Propionate + Salmeterol Xinafoate)
Arial Chemical_Formula
C25H37NO4
Arial RX_link
http://www.rxlist.com/cgi/generic/salmeterol.htm
Arial fda sheet
Arial msds (material safety sheet)
Arial Synthesis Reference
I. F. Skidmore et al., U.S. Pat. 4,992,474 (1991).
Arial Molecular Weight
415.566 g/mol
Arial Melting Point
75.5-76.5 oC
Arial H2O Solubility
Sparingly soluble
Arial State
Solid
Arial LogP
5.022
Arial Dosage Forms
Metered-dose (aerosol); Powder
Arial Indication
For the treatment of asthma and chronic obstructive pulmonary disease (COPD).
Arial Pharmacology
Salmeterol is a long acting beta2-adrenoceptor agonist (LABA), usually only prescribed for severe persistent asthma following previous treatment with a short-acting beta agonist such as salbutamol and is prescribed concurrently with a corticosteroid, such as beclometasone. The primary noticable difference of salmeterol to salbutamol is that the duration of action lasts approximately 12 hours in comparison with 4-6 hours of salbutamol. When used regularly every day as presecribed, inhaled salmeterol decreases the number and severity of asthma attacks. However, it is not for use for relieving an asthma attack that has already started. Inhaled salmeterol works like other beta 2-agonists, causing bronchodilatation by relaxing the smooth muscle in the airway so as to treat the exacerbation of asthma. Salmeterol is similar in action to formoterol, however formoterol has been demonstrated to have a faster onset of action than salmeterol as a result of a lower lipophilicity, and has also been demonstrated to be more potent - a 12 µg dose of formoterol has been demonstrated to be equivalent to a 50 µg dose of salmeterol.
Arial Absorption
Because of the small therapeutic dose, systemic levels of salmeterol are low or undetectable after inhalation of recommended doses.
Arial side effects and Toxicity
Symptoms of overdose include angina (chest pain), dizziness, dry mouth, fatigue, flu-like symptoms, headache, heart irregularities, high or low blood pressure, high blood sugar, insomnia, muscle cramps, nausea, nervousness, rapid heartbeat, seizures, and tremor. By the oral route, no deaths occurred in rats at 1,000 mg/kg (approximately 81,000 times the maximum recommended daily inhalation dose in adults and approximately 38,000 times the maximum recommended daily inhalation dose in children on a mg/m2 basis).
Arial Patient Information
PATIENT INFORMATION
Patients being treated with SEREVENT DISKUS should receive the following information and instructions. This information is
intended to aid them in the safe and effective use of this medication. It is not a disclosure of all possible adverse or
intended effects.
It is important that patients understand how to use the DISKUS appropriately and how to use SEREVENT DISKUS in relation to
other asthma or COPD medications they are taking. Patients should be given the following information:
1. The action of SEREVENT DISKUS may last up to 12 hours or longer. The recommended dosage (1 inhalation twice daily, morning
and evening) should not be exceeded.
2. Most patients are able to taste or feel a dose delivered from SEREVENT DISKUS. However, whether or not patients are able to
sense delivery of a dose, you should instruct them not to exceed the recommended dose of 1 inhalation twice daily, morning and
evening. You should instruct them to contact you or the pharmacist if they have questions.
3. SEREVENT DISKUS is not meant to relieve acute asthma or COPD symptoms and extra doses should not be used for that purpose.
Acute symptoms should be treated with an inhaled, short-acting bronchodilator (the physician should provide the patient with
such medication and instruct the patient in how it should be used).
4. Patients should not stop therapy with SEREVENT DISKUS for asthma or COPD without physician/provider guidance since symptoms
may worsen after discontinuation.
5. �When used for the treatment of EIB, 1 inhalation of SEREVENT DISKUS should be taken 30 minutes before exercise.
� Additional doses of SEREVENT should not be used for 12 hours.
� Patients who are receiving SEREVENT DISKUS twice daily should not use additional SEREVENT for prevention of EIB.
6. The physician should be notified immediately if any of the following situations occur, which may be a sign of seriously
worsening asthma or COPD:
� Decreasing effectiveness of inhaled, short-acting beta2-agonists
� Need for more inhalations than usual of inhaled, short-acting beta2-agonists
� Significant decrease in PEF or lung function as outlined by the physician
� Use of 4 or more inhalations per day of a short-acting beta2-agonist for 2 or more days consecutively
� Use of more than 1 canister (200 inhalations per canister) of an inhaled, short-acting beta2-agonist in an 8-week period.
7. SEREVENT DISKUS should not be used as a substitute for oral or inhaled corticosteroids. The dosage of these medications should
not be changed and they should not be stopped without consulting the physician, even if the patient feels better after initiating
treatment with SEREVENT DISKUS.
8. Patients should be cautioned regarding adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid
heart rate, tremor, or nervousness.
9. When patients are prescribed SEREVENT DISKUS, other medications for asthma and COPD should be used only as directed by the
physician.
10. SEREVENT DISKUS should not be used with a spacer device.
11. Patients who are pregnant or nursing should contact the physician about the use of SEREVENT DISKUS.
12. Effective and safe use of SEREVENT DISKUS includes an understanding of the way that it should be used:
� Never exhale into the DISKUS.
� Never attempt to take the DISKUS apart.
� Always activate and use the DISKUS in a level, horizontal position.
� Never wash the mouthpiece or any part of the DISKUS. KEEP IT DRY.
� Always keep the DISKUS in a dry place.
� Discard 6 weeks after removal from the moisture-protective foil overwrap pouch or after all blisters have been used (when the
dose indicator reads "0" ), whichever comes first.
13. For the proper use of SEREVENT DISKUS and to attain maximum benefit, the patient should read and follow carefully the Patient's
Instructions for Use accompanying the product.
Patients being treated with SEREVENT DISKUS should receive the following information and instructions. This information is
intended to aid them in the safe and effective use of this medication. It is not a disclosure of all possible adverse or
intended effects.
It is important that patients understand how to use the DISKUS appropriately and how to use SEREVENT DISKUS in relation to
other asthma or COPD medications they are taking. Patients should be given the following information:
1. The action of SEREVENT DISKUS may last up to 12 hours or longer. The recommended dosage (1 inhalation twice daily, morning
and evening) should not be exceeded.
2. Most patients are able to taste or feel a dose delivered from SEREVENT DISKUS. However, whether or not patients are able to
sense delivery of a dose, you should instruct them not to exceed the recommended dose of 1 inhalation twice daily, morning and
evening. You should instruct them to contact you or the pharmacist if they have questions.
3. SEREVENT DISKUS is not meant to relieve acute asthma or COPD symptoms and extra doses should not be used for that purpose.
Acute symptoms should be treated with an inhaled, short-acting bronchodilator (the physician should provide the patient with
such medication and instruct the patient in how it should be used).
4. Patients should not stop therapy with SEREVENT DISKUS for asthma or COPD without physician/provider guidance since symptoms
may worsen after discontinuation.
5. �When used for the treatment of EIB, 1 inhalation of SEREVENT DISKUS should be taken 30 minutes before exercise.
� Additional doses of SEREVENT should not be used for 12 hours.
� Patients who are receiving SEREVENT DISKUS twice daily should not use additional SEREVENT for prevention of EIB.
6. The physician should be notified immediately if any of the following situations occur, which may be a sign of seriously
worsening asthma or COPD:
� Decreasing effectiveness of inhaled, short-acting beta2-agonists
� Need for more inhalations than usual of inhaled, short-acting beta2-agonists
� Significant decrease in PEF or lung function as outlined by the physician
� Use of 4 or more inhalations per day of a short-acting beta2-agonist for 2 or more days consecutively
� Use of more than 1 canister (200 inhalations per canister) of an inhaled, short-acting beta2-agonist in an 8-week period.
7. SEREVENT DISKUS should not be used as a substitute for oral or inhaled corticosteroids. The dosage of these medications should
not be changed and they should not be stopped without consulting the physician, even if the patient feels better after initiating
treatment with SEREVENT DISKUS.
8. Patients should be cautioned regarding adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid
heart rate, tremor, or nervousness.
9. When patients are prescribed SEREVENT DISKUS, other medications for asthma and COPD should be used only as directed by the
physician.
10. SEREVENT DISKUS should not be used with a spacer device.
11. Patients who are pregnant or nursing should contact the physician about the use of SEREVENT DISKUS.
12. Effective and safe use of SEREVENT DISKUS includes an understanding of the way that it should be used:
� Never exhale into the DISKUS.
� Never attempt to take the DISKUS apart.
� Always activate and use the DISKUS in a level, horizontal position.
� Never wash the mouthpiece or any part of the DISKUS. KEEP IT DRY.
� Always keep the DISKUS in a dry place.
� Discard 6 weeks after removal from the moisture-protective foil overwrap pouch or after all blisters have been used (when the
dose indicator reads "0" ), whichever comes first.
13. For the proper use of SEREVENT DISKUS and to attain maximum benefit, the patient should read and follow carefully the Patient's
Instructions for Use accompanying the product.
Arial Organisms Affected
Humans and other mammals