Akbeta




Akbeta Brand names, Akbeta Analogs
Akbeta Brand Names Mixture
- No information avaliable
Akbeta Chemical_Formula
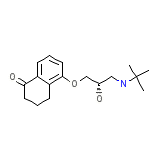
C17H25NO3
Akbeta RX_link
http://www.rxlist.com/cgi/generic/levobunolol.htm
Akbeta fda sheet
Akbeta msds (material safety sheet)
Akbeta Synthesis Reference
J. Shavel, Jr., S. Farber, U.S. Pat. 3,614,152 (1972)
Akbeta Molecular Weight
291.385 g/mol
Akbeta Melting Point
209-211oC
Akbeta H2O Solubility
No information avaliable
Akbeta State
Solid
Akbeta LogP
2.276
Akbeta Dosage Forms
Solution (sterile ophthalmic liquifilm)
Akbeta Indication
For lowering intraocular pressure and may be used in patients with chronic open-angle glaucoma or ocular hypertension
Akbeta Pharmacology
Levobunolol is an ophthalmic beta-blocker, equally effective at β(1)- and β(2)-receptor sites. The levorotatory isomer of bunolol, levobunolol is used for the treatment of IOP due to open-angle glaucoma or ocular hypertension.
Akbeta Absorption
80%
Akbeta side effects and Toxicity
Bradycardia, hypotension, bronchospasm, and acute cardiac failure, LD50=700 mg/kg (orally in rat).
Akbeta Patient Information
Description
--------------------------------------------------------------------------------
This medicine is used to manage glaucoma and to lower increased pressure in the eye
(intraocular pressure)
General Information
--------------------------------------------------------------------------------
This information is for educational purposes only. Not every known side effect,
adverse effect, or drug interaction is in this database. If you have questions about
your medicines, talk to your healthcare provider.
Proper use of this medicine
--------------------------------------------------------------------------------
Remove your contact lenses and leave them off for at least 15 minutes after you put
this medicine in your eyes. First, wash your hands. Tilt your head back and with your
index finger, form a pouch, by pulling your lid away from the eye. Gently place a drop
into the pouch and close your eyes. Immediately press your finger against the corner of
your eye to close off the tear duct, for 1 minute after you put the drop in your eye.
This will avoid excessive absorption into the body. Do not touch the applicator tip to
the eye or any other surface.
Missed Dose
--------------------------------------------------------------------------------
Take your next dose as soon as you remember. If it is time for your next dose, skip the
missed dose and go back to your regular schedule. Do not double doses.
Storage
--------------------------------------------------------------------------------
Protect from light. Store unopened bottle under refrigeration (36 to 46 degrees F).
Once opened may store at room temperature for 6 weeks.
Possible Side Effects
--------------------------------------------------------------------------------
burning of the eyes or irritation
blurred vision
growth of eyelashes
pigmentation of the eye (iridial)
This medicine can cause the following side effects:
Unreported side effects are possible, talk to your healthcare provider if any other
symptoms occur.
Warnings/Precautions
--------------------------------------------------------------------------------
Before using this medication, tell your doctor if you have:
asthma
diabets
heart disease
high blood pressure
kidney or liver disease
slow heart rate (bradycardia)
diabetes
thyroid disease
Do not use any eyedrop that is discolored or has particles in it.
Overdose
--------------------------------------------------------------------------------
Seek medical attention immediately. For non emergencies, contact your local or regional
poison control center
Drug Interactions
--------------------------------------------------------------------------------
Before using this medicine, tell your doctor if you are taking any of the following
medicines:
acetazolamide (Diamox)
dichlorphenamide (Daranide)
methazolamide (Neptazane)
propranolol (Inderal)
atenolol (Tenormin)
metoprolol (Lopressor; Toprol XL)
acebutolol (Sectral)
carvedilol (Coreg)
carteolol (Cartrol)
labetalol (Normodyne; Trandate)
pindolol (Visken)
sotalol (Betapace)
timolol (Blocadren)
Tell your doctor about any other mediciations that you are taking, including vitamins
and over-the-counter products.
Pregnancy/Nursing
--------------------------------------------------------------------------------
If you are pregnant or thinking of becoming pregnant talk to your doctor about the
benefits verses the risk of taking this medicine. It is unknown if this medicine is
excreted in breast milk. Talk to your doctor if you plan to breast feed while taking
this medicine.
More Information
--------------------------------------------------------------------------------
For more information about this medicine, talk to your healthcare provider.
--------------------------------------------------------------------------------
This medicine is used to manage glaucoma and to lower increased pressure in the eye
(intraocular pressure)
General Information
--------------------------------------------------------------------------------
This information is for educational purposes only. Not every known side effect,
adverse effect, or drug interaction is in this database. If you have questions about
your medicines, talk to your healthcare provider.
Proper use of this medicine
--------------------------------------------------------------------------------
Remove your contact lenses and leave them off for at least 15 minutes after you put
this medicine in your eyes. First, wash your hands. Tilt your head back and with your
index finger, form a pouch, by pulling your lid away from the eye. Gently place a drop
into the pouch and close your eyes. Immediately press your finger against the corner of
your eye to close off the tear duct, for 1 minute after you put the drop in your eye.
This will avoid excessive absorption into the body. Do not touch the applicator tip to
the eye or any other surface.
Missed Dose
--------------------------------------------------------------------------------
Take your next dose as soon as you remember. If it is time for your next dose, skip the
missed dose and go back to your regular schedule. Do not double doses.
Storage
--------------------------------------------------------------------------------
Protect from light. Store unopened bottle under refrigeration (36 to 46 degrees F).
Once opened may store at room temperature for 6 weeks.
Possible Side Effects
--------------------------------------------------------------------------------
burning of the eyes or irritation
blurred vision
growth of eyelashes
pigmentation of the eye (iridial)
This medicine can cause the following side effects:
Unreported side effects are possible, talk to your healthcare provider if any other
symptoms occur.
Warnings/Precautions
--------------------------------------------------------------------------------
Before using this medication, tell your doctor if you have:
asthma
diabets
heart disease
high blood pressure
kidney or liver disease
slow heart rate (bradycardia)
diabetes
thyroid disease
Do not use any eyedrop that is discolored or has particles in it.
Overdose
--------------------------------------------------------------------------------
Seek medical attention immediately. For non emergencies, contact your local or regional
poison control center
Drug Interactions
--------------------------------------------------------------------------------
Before using this medicine, tell your doctor if you are taking any of the following
medicines:
acetazolamide (Diamox)
dichlorphenamide (Daranide)
methazolamide (Neptazane)
propranolol (Inderal)
atenolol (Tenormin)
metoprolol (Lopressor; Toprol XL)
acebutolol (Sectral)
carvedilol (Coreg)
carteolol (Cartrol)
labetalol (Normodyne; Trandate)
pindolol (Visken)
sotalol (Betapace)
timolol (Blocadren)
Tell your doctor about any other mediciations that you are taking, including vitamins
and over-the-counter products.
Pregnancy/Nursing
--------------------------------------------------------------------------------
If you are pregnant or thinking of becoming pregnant talk to your doctor about the
benefits verses the risk of taking this medicine. It is unknown if this medicine is
excreted in breast milk. Talk to your doctor if you plan to breast feed while taking
this medicine.
More Information
--------------------------------------------------------------------------------
For more information about this medicine, talk to your healthcare provider.
Akbeta Organisms Affected
Humans and other mammals