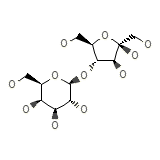
22,23-Dihydroxy-avermectin B




22,23-Dihydroxy-avermectin B Brand names, 22,23-Dihydroxy-avermectin B Analogs
22,23-Dihydroxy-avermectin B Brand Names Mixture
- No information avaliable
22,23-Dihydroxy-avermectin B Chemical_Formula
C12H22O11
22,23-Dihydroxy-avermectin B RX_link
http://www.rxlist.com/cgi/generic2/lactulose.htm
22,23-Dihydroxy-avermectin B fda sheet
22,23-Dihydroxy-avermectin B msds (material safety sheet)
22,23-Dihydroxy-avermectin B Synthesis Reference
No information avaliable
22,23-Dihydroxy-avermectin B Molecular Weight
342.296 g/mol
22,23-Dihydroxy-avermectin B Melting Point
173 - 178 oC
22,23-Dihydroxy-avermectin B H2O Solubility
764 mg/mL
22,23-Dihydroxy-avermectin B State
Solid
22,23-Dihydroxy-avermectin B LogP
-5.19
22,23-Dihydroxy-avermectin B Dosage Forms
Liquid; Powder; Solution; Syrup
22,23-Dihydroxy-avermectin B Indication
For the treatment of constipation and hepatic encephalopathy.
22,23-Dihydroxy-avermectin B Pharmacology
Therapeutically, lactulose has laxative and ammonia-detoxifying actions. In treating constipation lactulose metabolites draw water into the bowel, causing a cathartic effect through osmotic action.
22,23-Dihydroxy-avermectin B Absorption
Poorly absorbed from the gastrointestinal tract as no human enzyme that is capable of hydrolysis of this disaccharide is present in human gastrointestinal tissue.
22,23-Dihydroxy-avermectin B side effects and Toxicity
LD50=18.2 g/kg (oral, rat). Side effects include diarrhea and resultant dehydration.
22,23-Dihydroxy-avermectin B Patient Information
http://www.drugs.com/MTM/labetalol.html
22,23-Dihydroxy-avermectin B Organisms Affected
Humans and other mammals