VCZ




VCZ Brand names, VCZ Analogs
VCZ Brand Names Mixture
- No information avaliable
VCZ Chemical_Formula
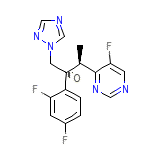
C16H14F3N5O
VCZ RX_link
http://www.rxlist.com/cgi/generic/vfend.htm
VCZ fda sheet
VCZ msds (material safety sheet)
VCZ Synthesis Reference
No information avaliable
VCZ Molecular Weight
349.311 g/mol
VCZ Melting Point
127 - 130 oC
VCZ H2O Solubility
Low
VCZ State
Solid
VCZ LogP
2.561
VCZ Dosage Forms
Tablet; Oral suspension; IV injection
VCZ Indication
For the treatment of esophageal candidiasis, invasive pulmonary aspergillosis, and serious fungal infections caused by Scedosporium apiospermum and Fusarium spp.
VCZ Pharmacology
Voriconazole is a triazole antifungal agent indicated for use in the treatment of fungal infections including invasive aspergillosis, esophageal candidiasis, and serious fungal infections caused by Scedosporium apiospermum (asexual form of Pseudallescheria boydii) and Fusarium spp. including Fusarium solani. Fungal plasma membranes are similar to mammalian plasma membranes, differing in having the nonpolar sterol ergosterol, rather than cholesterol, as the principal sterol. Membrane sterols such as ergosterol provide structure, modulation of membrane fluidity, and possibly control of some physiologic events. Voriconazole effects the formation of the fungal plasma membrane by indirectly inhibiting the biosynthesis of ergosterol. This results in plasma membrane permeability changes and inhibition of growth.
VCZ Absorption
The oral bioavailability is estimated to be 96% (CV 13%).
VCZ side effects and Toxicity
The minimum lethal oral dose in mice and rats was 300 mg/kg (equivalent to 4 and 7 times the recommended maintenance dose (RMD), based on body surface area). At this dose, clinical signs observed in both mice and rats included salivation, mydriasis, titubation (loss of balance while moving), depressed behavior, prostration, partially closed eyes, and dyspnea. Other signs in mice were convulsions, corneal opacification and swollen abdomen.
VCZ Patient Information
VCZ Organisms Affected
Yeast and other fungi