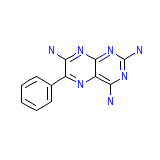
Teriam




Teriam Brand names, Teriam Analogs
Teriam Brand Names Mixture
- Apo Triazide Tab (Hydrochlorothiazide + Triamterene)
- Dyazide Tab (Hydrochlorothiazide + Triamterene)
- Novo-Triamzide (Hydrochlorothiazide + Triamterene)
- Nu-Triazide Tab 50 Mg/25 Mg (Hydrochlorothiazide + Triamterene)
- Penta-Triamterene HCTZ Tablets (Hydrochlorothiazide + Triamterene)
- Pro-Triazide (Hydrochlorothiazide + Triamterene)
- Riva-Zide 50/25mg Tablets (Hydrochlorothiazide + Triamterene)
Teriam Chemical_Formula
C12H11N7
Teriam RX_link
http://www.rxlist.com/cgi/generic/triamterine.htm
Teriam fda sheet
Teriam msds (material safety sheet)
Teriam Synthesis Reference
Spickett, Timmis, J. Chem.. Soc. 1954, 2887 (1958)
Teriam Molecular Weight
253.263 g/mol
Teriam Melting Point
316 oC
Teriam H2O Solubility
48.2 mg/L
Teriam State
Solid
Teriam LogP
-0.323
Teriam Dosage Forms
Tablet
Teriam Indication
For the treatment of edema associated with congestive heart failure, cirrhosis of the liver, and the nephrotic syndrome; also in steroid-induced edema, idiopathic edema, and edema due to secondary hyperaldosteronism.
Teriam Pharmacology
Triamterene, a relatively weak, potassium-sparing distal tubule diuretic and antihypertensive, is used in the management of hypokalemia. Triamterene is similar in action to amiloride but, unlike amiloride, increases the urinary excretion of magnesium.
Teriam Absorption
Rapidly absorbed, with somewhat less than 50% of the oral dose reaching the urine.
Teriam side effects and Toxicity
No information avaliable
Teriam Patient Information
Information for Patients
To help avoid stomach upset, it is recommended that the drug be taken after meals.
If a single daily dose is prescribed, it may be preferable to take it in the morning to minimize the effect of increased frequency of urination on nighttime sleep.
If a dose is missed, the patient should not take more than the prescribed dose at the next dosing interval.
Teriam Organisms Affected
Humans and other mammals