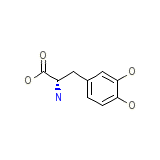
Cidandopa




Cidandopa Brand names, Cidandopa Analogs
- Atamet
- Bendopa
- Brocadopa
- Cidandopa
- DA
- DOPA
- Deadopa
- Dopaflex
- Dopaidan
- Dopal
- Dopal-Fher
- Dopalina
- Dopar
- Doparkine
- Doparl
- Dopasol
- Dopaston
- Dopastral
- Doprin
- Eldopal
- Eldopar
- Eldopatec
- Eurodopa
- Helfo-Dopa
- Insulamina
- L-DOPA
- L-Dihydroxyphenylalanine
- Laradopa
- Larodopa
- Ledopa
- Levedopa
- Levopa
- Maipedopa
- Parda
- Pardopa
- Prodopa
- Sinemet
- Sinemet CR
- Syndopa
- Veldopa
- Weldopa
Cidandopa Brand Names Mixture
- Apo-Levocarb CR Controlled-Release Tablets (carbidopa + levodopa)
- Dom-Levo-Carbidopa (carbidopa + levodopa)
- Novo-Levocarbidopa (carbidopa + levodopa)
- Pro-Lecarb-100/10 - Tab (carbidopa + levodopa)
- Pro-Lecarb-100/25 - Tab (carbidopa + levodopa)
- Prolopa Cap 50-12.5 (Benserazide + Levodopa)
- Ratio-Levodopa/Carbidopa (carbidopa + levodopa)
- Sinemet 100/25 (carbidopa + levodopa)
Cidandopa Chemical_Formula
C9H11NO4
Cidandopa RX_link
http://www.rxlist.com/cgi/generic3/stalevo.htm
Cidandopa fda sheet
Cidandopa msds (material safety sheet)
Cidandopa Synthesis Reference
Wasser, Lewandowski, Helv. Chem. Acta 4, 657(1921)
Cidandopa Molecular Weight
197.188 g/mol
Cidandopa Melting Point
276-278 oC
Cidandopa H2O Solubility
5000 mg/L
Cidandopa State
Solid
Cidandopa LogP
-2.244
Cidandopa Dosage Forms
Tablet (pink, scored, each containing levodopa 0.1 g)
Cidandopa Indication
For the treatment of idiopathic Parkinson's disease (Paralysis Agitans), postencephalitic parkinsonism, symptomatic parkinsonism which may follow injury to the nervous system by carbon monoxide intoxication, and manganese intoxication.
Cidandopa Pharmacology
Levodopa (L-dopa) is used to replace dopamine lost in Parkinson's disease because dopamine itself cannot cross the blood-brain barrier where its precursor can. However, L-DOPA is converted to dopamine in the periphery as well as in the CNS, so it is administered with a peripheral DDC (dopamine decarboxylase) inhibitor such as carbidopa, without which 90% is metabolised in the gut wall, and with a COMT inhibitor if possible; this prevents about a 5% loss. The form given therapeutically is therefore a prodrug which avoids decarboxylation in the stomach and periphery, can cross the blood-brain barrier, and once in the brain is converted to the neurotransmitter dopamine by the enzyme aromatic-L-amino-acid decarboxylase.
Cidandopa Absorption
Levodopa is rapidly absorbed from the proximal small intestine by the large neutral amino acid (LNAA) transport carrier system.
Cidandopa side effects and Toxicity
Oral, mouse: LD50 = 2363 mg/kg; Oral, rabbit: LD50 = 609 mg/kg; Oral, rat: LD50 = 1780 mg/kg.
Cidandopa Patient Information
Patient Instructions.
Stopping this medication suddenly can cause Parkinson��s
disease to worsen quickly. Report bothersome or unexpected side effects. Unless
prescribed, do not take levodopa in addition to this drug. Avoid pyridoxine (vitamin
B6) if you are taking levodopa alone, although it can be taken with carbidopa/
levodopa. Avoid high-protein meals for maximum absorption. If you are
taking the sustained-release tablet, swallow a whole or one-half tablet without
chewing or crushing it. Onset of effect of the first morning dose of the sustainedrelease
product could be delayed up to 1 hour compared with the quick-release
product. A dark color (red, brown, or black) might appear in saliva, urine, or
sweat and can stain clothing.
Stopping this medication suddenly can cause Parkinson��s
disease to worsen quickly. Report bothersome or unexpected side effects. Unless
prescribed, do not take levodopa in addition to this drug. Avoid pyridoxine (vitamin
B6) if you are taking levodopa alone, although it can be taken with carbidopa/
levodopa. Avoid high-protein meals for maximum absorption. If you are
taking the sustained-release tablet, swallow a whole or one-half tablet without
chewing or crushing it. Onset of effect of the first morning dose of the sustainedrelease
product could be delayed up to 1 hour compared with the quick-release
product. A dark color (red, brown, or black) might appear in saliva, urine, or
sweat and can stain clothing.
Cidandopa Organisms Affected
Humans and other mammals