Aldoclor-250




Aldoclor-250 Brand names, Aldoclor-250 Analogs
- AMD
- Aldoclor-150
- Aldoclor-250
- Aldomet
- Aldometil
- Aldomin
- Aldoril 15
- Aldoril 25
- Aldoril D30
- Aldoril D50
- Alpha medopa
- Alphamethyldopa
- Apo-Methyldopa
- Bayer 1440 L
- Baypresol
- Becanta
- Dopamet
- Dopamethyperpax
- Dopatec
- Dopegyt
- Grospisk
- Hyperpax
- Hypolag
- L-Methyl Dopa
- Medomet
- Medopa
- Medopal
- Medopren
- Methoplain
- Methyldopa anhydrous
- Methyldopate
- Methyldopate HCL
- Mk. b51
- Novomedopa
- Nu-Medopa
- Presinol
- Presolisin
- Sedometil
- Sembrina
Aldoclor-250 Brand Names Mixture
- No information avaliable
Aldoclor-250 Chemical_Formula
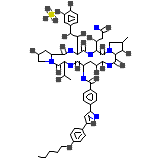
C56H71N9O23S
Aldoclor-250 RX_link
http://www.rxlist.com/cgi/generic4/mycamine.htm
Aldoclor-250 fda sheet
Aldoclor-250 msds (material safety sheet)
Aldoclor-250 Synthesis Reference
No information avaliable
Aldoclor-250 Molecular Weight
1270.28 g/mol
Aldoclor-250 Melting Point
No information avaliable
Aldoclor-250 H2O Solubility
Freely soluble as sodium salt (> 200mg/mL)
Aldoclor-250 State
Solid
Aldoclor-250 LogP
No information avaliable
Aldoclor-250 Dosage Forms
Powder for solution (for injection)
Aldoclor-250 Indication
For use in the treatment of patients with esophageal candidiasis and prophylaxis of Candida infections in patients undergoing hematopoietic stem cell transplantation.
Aldoclor-250 Pharmacology
Formerly known as FK463, micafungin is a novel antifungal agent. It is a glucan synthesis inhibitor of the echinocandin structural class. The U.S. Food and Drug Administration approved micafungin in March 2005. Micafungin inhibits an enzyme essential for fungal cell-wall synthesis and is fungicidal (lethal) for Candida. Micafungin can be used concomitantly with a variety of other drugs, including the HIV protease inhibitor ritonavir and the transplant medications cyclosporine and tacrolimus.
Aldoclor-250 Absorption
No information avaliable
Aldoclor-250 side effects and Toxicity
Intravenous LD50 in rats is 125mg/kg. In dogs it is >200mg/kg. No cases of overdosage have been reported. Repeated daily doses up to 8 mg/kg (maximum total dose of 896 mg) in adult patients have been administered in clinical trials with no reported dose-limiting toxicity. The minimum lethal dose is 125 mg/kg in rats, equivalent to 8.1 times the recommended human clinical dose for esophageal candidiasis based on body surface area comparisons.
Aldoclor-250 Patient Information
No information avaliable
Aldoclor-250 Organisms Affected
Aspergillis, Candida and other fungi